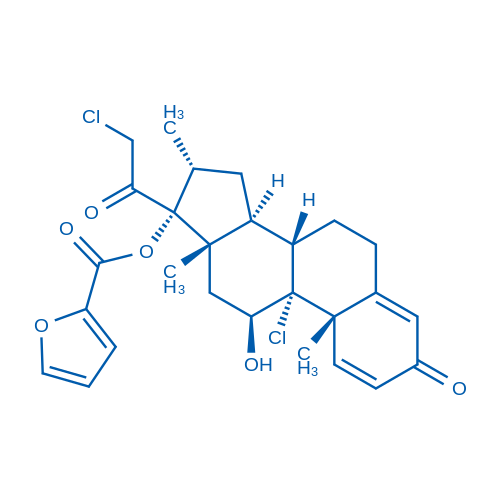
CAS No.: 83919-23-7
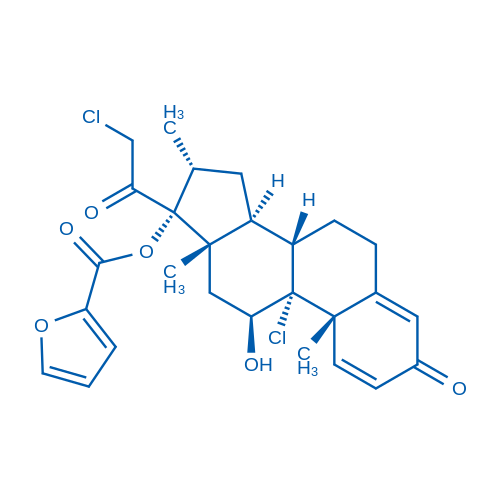
Mometasone furoate/糠酸莫美他松 Catalog No. CSN13867
Synonyms: Sch32088;Mometasone Furoate;ELOCON brand of mometasone furoate cream (0.1)
Mometasone furoate is a prodrug of mometasone with high affinity for glucocorticoid receptor.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00751075 Acute Rhinosinusitis Phase 3 Completed - - NCT01720797 - - Completed - - NCT01720797 Malocclusion Not Applicable Completed - United States, Florida ... more >> University of Florida, Department of Orthodontics Gainesville, Florida, United States, 32610 Collapse << - 更多
- 参考文献
- [1] Magari M, Ikeda M, et al. Suppressive effects of mometasone furoate on an antigen-specific IgE antibody response and production of IL-4 in mice. Immunopharmacol Immunotoxicol. 2006;28(3):491-500.
- [2] Tsumuro T, Ogawa M, et al. Effects of mometasone furoate on a rat allergic rhinitis model. Eur J Pharmacol. 2005 Nov 7;524(1-3):155-8.
- [3] Spada F, Barnes TM, Greive KA. Comparative safety and efficacy of topical mometasone furoate with other topical corticosteroids. Australas J Dermatol. 2018 Aug;59(3):e168-e174
- [4] Ho AY, Olm-Shipman M, Zhang Z, Siu CT, Wilgucki M, Phung A, Arnold BB, Porinchak M, Lacouture M, McCormick B, Powell SN, Gelblum DY. A Randomized Trial of Mometasone Furoate 0.1% to Reduce High-Grade Acute Radiation Dermatitis in Breast Cancer Patients Receiving Postmastectomy Radiation. Int J Radiat Oncol Biol Phys. 2018 Jun 1;101(2):325-333
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 83919-23-7 | 储存条件 |
|
|||||||
| 分子式 | C27H30Cl2O6 | 运输 | 蓝冰 | |||||||
| 分子量 | 521.43 | 别名 | Sch32088;Mometasone Furoate;ELOCON brand of mometasone furoate cream (0.1);糠酸莫米松 | |||||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00751075 | Acute Rhinosinusitis | Phase 3 | Completed | - | - |
| NCT01720797 | - | - | Completed | - | - |
| NCT01720797 | Malocclusion | Not Applicable | Completed | - | United States, Florida ... more >> University of Florida, Department of Orthodontics Gainesville, Florida, United States, 32610 Collapse << |
| NCT00711165 | Asthma | Phase 4 | Completed | - | United States, Nebraska ... more >> Creighton University Medical Center Omaha, Nebraska, United States, 68131 Collapse << |
| NCT00750750 | Acute Rhinosinusitis | Phase 2 | Completed | - | - |
| NCT00224523 | Rhinitis, Allergic, Perennial | Phase 3 | Completed | - | Belgium ... more >> GSK Investigational Site Leuven, Belgium, 3000 Netherlands GSK Investigational Site Amsterdam, Netherlands, 1105 AZ GSK Investigational Site Den Haag, Netherlands, 2566 MJ GSK Investigational Site Nijmegen, Netherlands, 6525 EX Collapse << |
| NCT01038427 | Seasonal Allergic Rhinitis | Phase 3 | Completed | - | United States, Texas ... more >> Austin, Texas, United States, 78731/50/59 Kerrville, Texas, United States, 78028 Live Oak, Texas, United States, 78233 New Braunfels, Texas, United States, 78310 Sylvana Research San Antonio, Texas, United States, 78229 San Antonio, Texas, United States, 78229 Waco, Texas, United States, 76708 Collapse << |
| NCT03401229 | Nasal Polyposis | Phase 3 | Recruiting | November 4, 2020 | - |
| NCT03060057 | - | - | Recruiting | March 30, 2024 | United States, Florida ... more >> Coastal Orthopedics Recruiting Bradenton, Florida, United States, 34209 Contact: Christine Scheuerman, RN, BSN 941-782-1353 ext 3450 cscheuerman@coastalorthopedics.com Principal Investigator: David Cashen, MD Sub-Investigator: Alan Valadie, MD Sub-Investigator: Arthur Valadie, MD Sub-Investigator: Daniel Lamar, MD Jacksonville Orthopaedic Institute Recruiting Jacksonville, Florida, United States, 32204 Contact: Carol Collins 904-388-1400 Principal Investigator: Stanton Longenecker, MD Orthopedic Center of Vero Beach Recruiting Vero Beach, Florida, United States, 32960 Contact: Nicki Allen 772-778-2009 Principal Investigator: Richard Steinfeld, MD United States, Maryland Rubin Institute for Advanced Orthopedics Not yet recruiting Baltimore, Maryland, United States, 21215 Contact: Ashwin Mahajan 410-601-8500 Sub-Investigator: Ronald Delanois, MD Principal Investigator: James Nace, MD Collapse << |
| NCT00967967 | Asthma Rhinit... more >>is Collapse << | Not Applicable | Suspended(Awaiting results fro... more >>m sub analysis to decide if study continuesé) Collapse << | September 2014 | Canada, Quebec ... more >> Institut universitaire de cardiologie et de pneumologie de Québec Québec, Quebec, Canada, G1V 4G5 Collapse << |
| NCT00423176 | - | - | Terminated(Based on business p... more >>riorities. Not related to any safety or efficacy issue & took place before data were unblinded or analyzed) Collapse << | - | - |
| NCT00423176 | Sinusitis | Phase 3 | Terminated(Based on business p... more >>riorities. Not related to any safety or efficacy issue & took place before data were unblinded or analyzed) Collapse << | - | - |
| NCT02291549 | Chronic Sinusitis ... more >> Nasal Polyposis Collapse << | Phase 3 | Completed | - | - |
| NCT00424008 | Asthma | Phase 3 | Completed | - | - |
| NCT00424008 | - | - | Completed | - | - |
| NCT02109185 | Treatment of the Signs and Sym... more >>ptoms of Seasonal Allergic Rhinitis Collapse << | Phase 1 | Completed | - | - |
| NCT01732536 | Chronic Sinusitis ... more >> Nasal Polyps Collapse << | Phase 2 Phase 3 | Completed | - | United States, California ... more >> California Sinus Centers Atherton, California, United States, 94027 Cedars-Sinai Medical Center, Sinus Center of Excellence Los Angeles, California, United States, 90048 Sacramento Ear, Nose and Throat Sacramento, California, United States, 95815 United States, Colorado Colorado Ear, Nose, Throat & Allergy Colorado Springs, Colorado, United States, 80909 United States, Florida South Florida ENT Associates Miami, Florida, United States, 33176 United States, Georgia ENT of Georgia Atlanta, Georgia, United States, 30342 United States, Illinois Northwestern University, Department of Otolaryngology-Head & Neck Surgery Chicago, Illinois, United States, 60611 United States, Kentucky Advanced ENT & Allergy Louisville, Kentucky, United States, 40207 United States, New Jersey Summit Medical Group Berkeley Heights, New Jersey, United States, 07922 United States, New York Albany ENT & Allergy Services Albany, New York, United States, 12206 ENT and Allergy Associates Lake Success, New York, United States, 11042 United States, North Carolina Piedmont Ear, Nose & Throat Associates Winston-Salem, North Carolina, United States, 27103 United States, Oregon Oregon Health and Science University-Dept. of Otolaryngology Head and Neck Surgery Portland, Oregon, United States, 97239 United States, Pennsylvania Bethlehem ENT Specialty Physicians Associates Bethlehem, Pennsylvania, United States, 18017 University of Pennsylvania, Dept of Otolaryngology-Head & Neck Surgery Philadelphia, Pennsylvania, United States, 19104 United States, South Carolina Medical University of South Carolina Charleston, South Carolina, United States, 29425 United States, Utah Intermountain Ear, Nose & Throat Center Salt Lake City, Utah, United States, 84102 United States, Virginia Eastern Virginia Medical School Department of Otolaryngology Norfolk, Virginia, United States, 23507 United States, Wisconsin Medical College of Wisconsin Milwaukee, Wisconsin, United States, 53266 Collapse << |
| NCT01732536 | - | - | Completed | - | - |
| NCT00662883 | Healthy | Phase 1 | Completed | - | United States, Maryland ... more >> Baltimore, Maryland, United States, 21225 Collapse << |
| NCT02291549 | - | - | Completed | - | - |
| NCT02628886 | Pneumonia | Phase 3 | Recruiting | November 2019 | Gambia ... more >> Sukuta Health Centre Recruiting Banjul, Gambia Contact: Francis Oko, MB +2204495443 ext 3035 foko@mrc.gm Contact: Ed Clarke, MB +2204495443 ext 4014 eclarke@mrc.gm Collapse << |
| NCT00379288 | Asthma | Phase 3 | Completed | - | - |
| NCT02554786 | Asthma | Phase 3 | Recruiting | June 24, 2019 | - |
| NCT00379288 | - | - | Completed | - | - |
| NCT00418509 | Healthy | Phase 1 | Completed | - | United Kingdom ... more >> Novartis Horsham, United Kingdom Collapse << |
| NCT01353599 | Spinal Cord Injury | Phase 1 | Unknown | - | United States, New Jersey ... more >> Kessler Institute for Rehabilitation West Orange, New Jersey, United States, 07052 United States, New York James J. Peters VA Medical Center Bronx, New York, United States, 10468 Collapse << |
| NCT00552110 | Seasonal Allergic Rhinitis | Phase 2 | Completed | - | - |
| NCT03444506 | Seasonal Allergic Rhinitis | Phase 2 | Completed | - | Canada, Ontario ... more >> Glenmark Investigational Site 1 Mississauga, Ontario, Canada, L4W 1V7 Collapse << |
| NCT00552110 | - | - | Completed | - | - |
| NCT01258803 | Asthma | Phase 2 | Completed | - | - |
| NCT01258803 | - | - | Completed | - | - |
| NCT01199757 | - | - | Completed | - | - |
| NCT00763529 | Psoriasis | Phase 4 | Completed | - | - |
| NCT00383435 | - | - | Completed | - | - |
| NCT00383721 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 3 | Completed | - | - |
| NCT00383721 | - | - | Completed | - | - |
| NCT03705793 | Chronic Rhinosinusitis (Diagno... more >>sis) Allergic Rhinosinusitis Chronic Eosinophilic Rhinosinusitis Collapse << | Phase 4 | Not yet recruiting | July 2019 | - |
| NCT00383435 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 3 | Completed | - | - |
| NCT00783237 | Allergic Rhinitis | Phase 4 | Completed | - | - |
| NCT00635882 | Asthma Airway... more >> Inflammation Collapse << | Phase 2 | Completed | - | - |
| NCT03637998 | Chronic Low Back Pain ... more >> Self-Management Collapse << | Not Applicable | Recruiting | June 2020 | United States, Connecticut ... more >> University of Connecticut Recruiting Storrs, Connecticut, United States, 06269 Contact: Kyounghae Kim, PhD, RN 860-486-5915 kyounghae.kim@uconn.edu Contact: Angela Starkweather, PhD, RN 860-486-0549 angela.starkweather@uconn.edu Principal Investigator: Kyounghae Kim, PhD, RN Collapse << |
| NCT01671852 | Obstructive Sleep Apnea (Mild,... more >> Moderate, Severe) as Per Polysomnography Collapse << | Phase 3 | Withdrawn(Could not obtain fun... more >>ding.) Collapse << | - | Canada, British Columbia ... more >> BC Women's and Children's Hospital Vancouver, British Columbia, Canada, V6H 3N1 Collapse << |
| NCT00783224 | Perennial Allergic Rhinitis | Phase 3 | Completed | - | - |
| NCT00635882 | - | - | Completed | - | - |
| NCT02741271 | Asthma | Phase 3 | Completed | - | - |
| NCT00783224 | - | - | Completed | - | - |
| NCT00394355 | Asthma | Phase 4 | Completed | - | - |
| NCT00383240 | Asthma | Phase 3 | Completed | - | - |
| NCT00383552 | Asthma | Phase 3 | Completed | - | - |
| NCT00394355 | - | - | Completed | - | - |
| NCT00383552 | - | - | Completed | - | - |
| NCT02397915 | Rhinitis, Allergic, Perennial ... more >>and Seasonal Collapse << | Phase 4 | Completed | - | Argentina ... more >> GSK Investigational Site Buenos Aires, Argentina, C1425BEN GSK Investigational Site Ciudad Autónoma de Buenos Aires, Argentina, C1426ABP GSK Investigational Site Mendoza, Argentina, 5500 GSK Investigational Site Mendoza, Argentina, M5500CCG Australia, New South Wales GSK Investigational Site Coffs Harbour, New South Wales, Australia, 2450 GSK Investigational Site Maroubra, New South Wales, Australia, 2035 Australia, Western Australia GSK Investigational Site Murdoch, Western Australia, Australia, 6150 Korea, Republic of GSK Investigational Site Incheon, Korea, Republic of, 405-760 GSK Investigational Site Seongnam-si, Gyeonggi-do, Korea, Republic of, 463-707 GSK Investigational Site Seoul, Korea, Republic of, 08308 Russian Federation GSK Investigational Site Moscow, Russian Federation, 123182 GSK Investigational Site Stavropol, Russian Federation, 355030 Collapse << |
| NCT02397915 | - | - | Completed | - | - |
| NCT02687438 | Chronic Sinusitis | Not Applicable | Completed | - | United States, New York ... more >> ENT and Allergy Associates, LLP Lake Success, New York, United States, 11042 ENT and Allergy Associates, LLP New York, New York, United States, 10016 ENT and Allergy Associates, LLP Port Jefferson, New York, United States, 11777 ENT and Allergy Associates, LLP White Plains, New York, United States, 10601 Collapse << |
| NCT02559440 | Adenoidal Hypertrophy | Phase 4 | Completed | - | - |
| NCT00604500 | Asthma COPD | Phase 3 | Completed | - | - |
| NCT00383240 | - | - | Completed | - | - |
| NCT00604500 | - | - | Completed | - | - |
| NCT00746330 | - | - | Completed | - | - |
| NCT00521599 | Asthma | Phase 4 | Completed | - | - |
| NCT00521599 | - | - | Completed | - | - |
| NCT01471340 | Asthma | Phase 4 | Completed | - | - |
| NCT00746330 | Asthma | Phase 2 | Completed | - | United States, Alabama ... more >> Novartis Investigative Site Mobile, Alabama, United States, 36608 United States, California Novartis Investigative Site Huntington Beach, California, United States, 92647 Novartis Investigative Site Orange, California, United States, 92868 United States, Florida Novartis Investigative Site Hialeah, Florida, United States, 33012 Novartis Investigative Site Pensacola, Florida, United States, 32503 United States, Oklahoma Novartis Investigative Site Oklahoma City, Oklahoma, United States, 73112 Colombia Novartis Investigative Site Barranquilla, Colombia Novartis Investigative Site Bogota, Colombia Peru Novartis Investigative Site Lima, Peru Collapse << |
| NCT01566149 | Asthma | Phase 3 | Completed | - | - |
| NCT01523236 | Seasonal Allergic Rhinitis | Phase 1 Phase 2 | Completed | - | - |
| NCT01471340 | - | - | Completed | - | - |
| NCT01386125 | Nasal Polyps | Phase 3 | Completed | - | - |
| NCT02967731 | Chronic Sinusitis | Phase 1 | Active, not recruiting | June 2018 | Australia ... more >> The Queen Elizabeth Hospital Adelaide, Australia Royal Brisbane and Women's Hospital Brisbane, Australia Monash Medical Center Melbourne, Australia New Zealand University of Auckland Auckland, New Zealand Collapse << |
| NCT00779545 | Rhinitis, Allergic, Perennial | Phase 2 | Completed | - | - |
| NCT01386125 | - | - | Completed | - | - |
| NCT01502371 | Asthma | Phase 2 | Completed | - | - |
| NCT01566149 | - | - | Completed | - | - |
| NCT00728416 | - | - | Completed | - | - |
| NCT00733005 | Allergic Rhinitis | Phase 3 | Completed | - | - |
| NCT00731185 | Nasal Polyps | Phase 3 | Completed | - | - |
| NCT01026870 | Asthma | Phase 3 | Withdrawn | December 2013 | - |
| NCT00728416 | Allergic Rhinitis | Phase 3 | Completed | - | - |
| NCT01502371 | - | - | Completed | - | - |
| NCT02951338 | Stroke | Not Applicable | Enrolling by invitation | June 2020 | Canada, Ontario ... more >> Hamilton Health Sciences Hamilton, Ontario, Canada, L8L 2X2 St. Joseph's Care Group Thunder Bay, Ontario, Canada, P7B 5G7 Sunnybrook Research Institute Toronto, Ontario, Canada, M4N 3M5 West Park Healthcare Centre Toronto, Ontario, Canada, M5G 1V7 Toronto Rehabilitation Institute - UHN Toronto, Ontario, Canada, M5G 2A2 Collapse << |
| NCT00732381 | - | - | Completed | - | - |
| NCT00733005 | - | - | Completed | - | - |
| NCT00732381 | Allergic Rhinitis | Phase 3 | Completed | - | - |
| NCT00442117 | - | - | Completed | - | - |
| NCT00903721 | - | - | Completed | - | - |
| NCT00442117 | Asthma | Phase 3 | Completed | - | - |
| NCT00732368 | Perennial Allergic Rhinitis | Phase 3 | Completed | - | - |
| NCT00438659 | Breast Cancer ... more >> Dermatologic Complications Radiation Toxicity Skin Reactions Secondary to Radiation Therapy Collapse << | Phase 3 | Completed | - | - |
| NCT00605306 | - | - | Completed | - | - |
| NCT00783458 | Allergic Rhinitis | Phase 4 | Completed | - | - |
| NCT00381485 | Asthma | Phase 3 | Completed | - | - |
| NCT00817050 | Allergic Rhinitis | Phase 4 | Completed | - | - |
| NCT00438659 | - | - | Completed | - | - |
| NCT00834119 | Rhinitis, Allergic | Phase 4 | Completed | - | - |
| NCT00605306 | Asthma | Phase 2 | Completed | - | France ... more >> Novartis Investigator Site Neuil, France Novartis Investigator Site Paris, France Novartis Investigator Site Poitiers, France Collapse << |
| NCT00491504 | Seasonal Allergic Rhinitis | Phase 4 | Completed | - | - |
| NCT00491374 | Perennial Allergic Rhinitis | Phase 4 | Terminated(Very poor enrollmen... more >>t) Collapse << | - | - |
| NCT00381485 | - | - | Completed | - | - |
| NCT00491504 | - | - | Completed | - | - |
| NCT00442351 | Asthma | Phase 4 | Terminated(Slow Enrollment) | - | - |
| NCT00491374 | - | - | Terminated(Very poor enrollmen... more >>t) Collapse << | - | - |
| NCT00442351 | - | - | Terminated(Slow Enrollment) | - | - |
| NCT01894503 | Chronic Sinusitis | Phase 2 | Completed | - | United States, California ... more >> Sacramento ENT Sacramento, California, United States, 95815 Collapse << |
| NCT00805155 | - | - | Completed | - | - |
| NCT01894503 | - | - | Completed | - | - |
| NCT00552032 | Adenoids Hypertrophy | Phase 3 | Completed | - | - |
| NCT00453063 | Seasonal Allergic Rhinitis | Phase 3 | Completed | - | - |
| NCT02880514 | Chronic Sinusitis | Not Applicable | Completed | - | United States, California ... more >> Sacramento ENT Sacramento, California, United States, 95815 United States, Florida ENT Assoicates of South Florida Boca Raton, Florida, United States, 33487 United States, Georgia ENT of Georgia Atlanta, Georgia, United States, 30342 United States, Kentucky Advanced ENT and Allergy Louisville, Kentucky, United States, 40207 United States, Louisiana Associated Surgical Specialists Covington, Louisiana, United States, 70433 United States, Missouri St. Luke's ENT Specialists Kansas City, Missouri, United States, 64111 United States, New Mexico BreatheAmerica of Albuquerque Albuquerque, New Mexico, United States, 87109 United States, New York Madison ENT New York, New York, United States, 10016 United States, Ohio Ohio Sinus Institute Dublin, Ohio, United States, 43016 Collapse << |
| NCT00358527 | Seasonal Allergic Rhinitis | Phase 4 | Completed | - | - |
| NCT00358527 | - | - | Completed | - | - |
| NCT00378378 | Nasal Polyps | Phase 3 | Completed | - | - |
| NCT00552032 | - | - | Completed | - | - |
| NCT00359216 | Perennial Allergic Rhinitis ... more >> Obstructive Sleep Apnea Sleep Disorder Collapse << | Phase 4 | Completed | - | - |
| NCT00378378 | - | - | Completed | - | - |
| NCT00453063 | - | - | Completed | - | - |
| NCT01555151 | Asthma | Phase 2 | Completed | - | - |
| NCT02880514 | - | - | Completed | - | - |
| NCT00359216 | - | - | Completed | - | - |
| NCT02266810 | Chronic Sinusitis | Phase 3 | Completed | - | United States, California ... more >> Sacramento Ear, Nose and Throat Sacramento, California, United States, 95815 Breathe Clear Institute of Sinus and Allergy Relief Torrance, California, United States, 90503 United States, Connecticut The Connecticut Center for Advanced ENT Care Norwalk, Connecticut, United States, 06851 United States, District of Columbia George Washington University Medical Faculty Associates Washington, District of Columbia, United States, 20006 United States, Georgia ENT of Georgia Atlanta, Georgia, United States, 30342 United States, Kentucky Advanced ENT and Allergy Louisville, Kentucky, United States, 40207 United States, New York Albany ENT and Allergy Albany, New York, United States, 12206 United States, Oregon Oregon Health and Science University Portland, Oregon, United States, 97239 United States, Texas University of Texas Health Science Center at Houston Houston, Texas, United States, 77030 United States, Virginia East Virginia Medical School Norfolk, Virginia, United States, 23507 Collapse << |
| NCT01856543 | Invasive Breast Cancer | Phase 3 | Completed | - | United States, New Jersey ... more >> Memorial Sloan Kettering Cancer Center at Basking Ridge Basking Ridge, New Jersey, United States, 07939 United States, New York Memorial Sloan Kettering Cancer Center @ Commack Commack, New York, United States, 11725 Memorial Sloan Kettering West Harrison Harrison, New York, United States, 10604 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Memorial Sloan Kettering Cancer Center at Mercy Medical Center Rockville Centre, New York, United States, 11570 Memorial Sloan Kettering Cancer Center at Phelps Memorial Hospital Center Sleepy Hollow, New York, United States, 10591 Collapse << |
| NCT00468312 | Seasonal Allergic Rhinitis | Phase 3 | Completed | - | - |
| NCT01555151 | - | - | Completed | - | - |
| NCT00468312 | - | - | Completed | - | - |
| NCT02266810 | - | - | Completed | - | - |
| NCT00556673 | Asthma | Phase 2 | Completed | - | France ... more >> Novartis Investigator Site Poitiers, France, 86000 Germany Novartis Investigator Site Berlin, Germany, 14050 Collapse << |
| NCT00779740 | Rhinitis, Allergic, Perennial | Phase 3 | Completed | - | - |
| NCT00835094 | Asthma | Phase 4 | Completed | - | - |
| NCT00553891 | Nasal Obstruction ... more >> Adenoids Hypertrophy Adenoidectomy Collapse << | Phase 4 | Terminated(No enrollment becau... more >>se of war in the study country.) Collapse << | - | - |
| NCT01165424 | - | - | Completed | - | - |
| NCT01165424 | Rhinitis, Allergic, Perennial | Phase 3 | Completed | - | - |
| NCT00556673 | - | - | Completed | - | - |
| NCT01135134 | Rhinitis, Allergic, Perennial | Phase 3 | Completed | - | - |
| NCT01616160 | - | - | Terminated(Inability to recrui... more >>t additional subjects.) Collapse << | - | - |
| NCT01135134 | - | - | Completed | - | - |
| NCT03534362 | Stent Sinusit... more >>is, Frontal Collapse << | Not Applicable | Enrolling by invitation | December 31, 2022 | United States, Missouri ... more >> Saint Louis Universtiy Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT01098071 | Adenoids | Phase 4 | Completed | - | - |
| NCT02125253 | Seasonal Allergic Rhinitis | Phase 3 | Completed | - | United States, Texas ... more >> Isis Clinical Research Austin, Texas, United States, 78731 Austin Clinical Research Austin, Texas, United States, 78750 Sirius Clinical Research Austin, Texas, United States, 78759 Kerrville Research Associates Kerrville, Texas, United States, 78028 Central Texas Health Research New Braunfels, Texas, United States, 78130 Allergy & Asthma Research Center San Antonio, Texas, United States, 78229 Biogenics Research Institute San Antonio, Texas, United States, 78229 Diagnostics Research Group San Antonio, Texas, United States, 78229 Sylvania Research San Antonio, Texas, United States, 78229 Allergy & Asthma Care of Waco Waco, Texas, United States, 76708 Collapse << |
| NCT01616160 | Nasal Polyps | Phase 4 | Terminated(Inability to recrui... more >>t additional subjects.) Collapse << | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT01098071 | - | - | Completed | - | - |
| NCT00599027 | Allergic Rhinitis ... more >> Asthma Collapse << | Phase 3 | Completed | - | - |
| NCT01470053 | Perennial Allergic Rhinitis | Phase 3 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT01228656 | Plaque Psoriasis | Phase 2 | Suspended | October 2011 | - |
| NCT01333410 | Vitiligo | Phase 4 | Unknown | December 2013 | Thailand ... more >> Department of Dermatology, Faculty of Medicine Siriraj Hospital Bangkok, Thailand Collapse << |
| NCT00599027 | - | - | Completed | - | - |
| NCT02113267 | Eosinophilic Esophagitis | Phase 2 | Terminated(Combination slow re... more >>cruitment, short shelf life for placebo and insufficient funding for further drug production (probably anyway sufficient number included).) Collapse << | - | Sweden ... more >> ENT dept, NÄL Hospital Trollhättan, Sweden, SE46185 Collapse << |
| NCT02495064 | Early Radiation Dermatitis | Phase 4 | Unknown | December 2016 | China, Sichuan ... more >> oncology department of Mian yang central Hosptial Recruiting Mianyang, Sichuan, China, 0816 Contact: Liao Yao, Resident 0086-13778106466 425838503@qq.com Contact: Du x bo, Chief 0086-13550822229 duxiaobo2005@126.com Collapse << |
| NCT00687531 | Asthma | Phase 4 | Terminated | - | - |
| NCT02588326 | Hypersensitivity | Phase 1 | Recruiting | July 2019 | United States, Florida ... more >> Department of Pharmaceutics, University of Florida Recruiting Gainesville, Florida, United States, 32610 Sub-Investigator: Guenther Hochhaus, PhD Principal Investigator: Juergen Bulitta, PhD Collapse << |
| NCT00687531 | - | - | Terminated | - | - |
| NCT02110654 | Sinusitis Nas... more >>al Polyps Asthma Collapse << | Phase 4 | Unknown | June 2016 | China, Guangdong ... more >> the first affiliated hospital, Sun Yat-sen University Not yet recruiting Guangzhou, Guangdong, China, 510080 Contact: Fenghong Chen, doctor 02086013560170816 chfhong@mail.sysu.edu.cn Principal Investigator: jianbo Shi, doctor Collapse << |
| NCT02228720 | Chronic Sinusitis | Not Applicable | Completed | - | United States, Florida ... more >> South Florida ENT Associates Miami, Florida, United States, 33176 United States, Ohio Ohio Sinus Institute Dublin, Ohio, United States, 43016 Collapse << |
| NCT02228720 | - | - | Completed | - | - |
| NCT00727571 | - | - | Completed | - | - |
| NCT00576069 | - | - | Unknown | June 2018 | United States, California ... more >> Arthur F Gelb Medical Corporation Recruiting Lakewood, California, United States, 90712 Principal Investigator: Arthur F Gelb, MD Collapse << |
| NCT02415179 | Asthma | Not Applicable | Completed | - | Thailand ... more >> Faculty of Medicine, Ramathibodi Hospital Bangkok, Thailand Collapse << |
| NCT01204047 | - | - | Completed | - | United States, Florida ... more >> The Miami Project to Cure Paralysis Miami, Florida, United States, 33136 Collapse << |
| NCT00727571 | - | - | Completed | - | - |
| NCT01854047 | Asthma | Phase 2 | Completed | - | - |
| NCT01676415 | Chronic Rhinosinusitis Without... more >> Nasal Polyps Collapse << | Phase 4 | Terminated(participants are no... more >> longer receiving intervention due to clinical logistics) Collapse << | - | United States, Illinois ... more >> : Northwestern Medical Faculty Foundation (NMFF) Sinus and Allergy Center Chicago, Illinois, United States, 60611 Collapse << |
| NCT03683758 | Soccer Exerci... more >>se Athletic Performance Collapse << | Not Applicable | Active, not recruiting | November 11, 2018 | Canada ... more >> Windsor Bubble North Vancouver, Canada Collapse << |
| NCT01854047 | - | - | Completed | - | - |
| NCT03240770 | Malocclusion | Not Applicable | Completed | - | United States, New Jersey ... more >> CTOR Hoboken, New Jersey, United States, 07030 Orthodontia Studio Hoboken, New Jersey, United States, 07030 United States, New York House of Orthodontia Brooklyn, New York, United States, 11201 United States, Texas Atique Orthodontics San Antonio, Texas, United States, 78259 Collapse << |
| NCT02154334 | Allergic Rhinitis | Phase 1 | Completed | - | Germany ... more >> Hannover, Germany Collapse << |
| NCT02805907 | - | - | Completed | - | - |
| NCT03727893 | Spinal Cord Injuries | Not Applicable | Recruiting | July 31, 2019 | United States, Missouri ... more >> Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63108 Contact: Kimberly A Walker, OTD 314-273-7010 walker.k@wustl.edu Contact: Kerri A Morgan, PhD 314-286-1659 morgank@wustl.edu Collapse << |
| NCT03193229 | Pre Diabetes ... more >>Obesity Collapse << | Not Applicable | Completed | - | United States, Iowa ... more >> Signal Center Innovation Lab Coralville, Iowa, United States, 52241 Collapse << |
| NCT01676415 | - | - | Terminated(participants are no... more >> longer receiving intervention due to clinical logistics) Collapse << | - | - |
| NCT01184729 | - | - | Completed | - | United States, Florida ... more >> The Miami Project to Cure Paralysis Miami, Florida, United States, 33136 Collapse << |
| NCT01639040 | Atopic Dermatitis | Phase 2 | Completed | - | Germany ... more >> Berlin, Germany Dresden, Germany Duelmen, Germany Frankfurt, Germany Gera, Germany Langenau, Germany Munster, Germany Hungary Szeged-Hungary, Hungary Szolnok, Hungary Poland Gdansk, Poland Lodz, Poland Lublin, Poland Warszawa, Poland Collapse << |
| NCT02805907 | Asthma, Bronchial | Phase 4 | Completed | - | Spain ... more >> Servicio Murciano de Salud Murcia, Spain, 30008 Collapse << |
| NCT01639040 | - | - | Completed | - | - |
| NCT03746665 | Meningitis Me... more >>ningococcal Collapse << | Phase 3 | Not yet recruiting | November 2020 | - |
| NCT02709538 | Perennial Allergic Rhinitis | Phase 3 | Completed | - | - |
| NCT03720054 | Veterans Obes... more >>ity Overweight Collapse << | Not Applicable | Recruiting | December 2018 | United States, Iowa ... more >> VA Health Care System Recruiting Iowa City, Iowa, United States, 52246 Contact: Jessica Trepanier, MBA 319-338-0581 ext 3538 Jessica.Trepanier@va.gov Principal Investigator: Philip Polgreen, MD Collapse << |
| NCT02709538 | - | - | Completed | - | - |
| NCT00845481 | - | - | Completed | - | - |
| NCT03469414 | - | - | Recruiting | December 31, 2018 | United States, Minnesota ... more >> Courage Kenny Rehabilitation Institute Recruiting Golden Valley, Minnesota, United States, 55422 Collapse << |
| NCT02592434 | Juvenile Idiopathic Arthritis | Phase 3 | Recruiting | May 21, 2019 | - |
| NCT00845481 | Psoriasis Vulgaris | Phase 2 | Completed | - | France ... more >> LEO Pharma site St Quentin Yvelines Cedex, France, 78054 Collapse << |
| NCT01900912 | Diarrhea Resp... more >>iratory Infection Collapse << | Not Applicable | Completed | - | Mali ... more >> Rural Communities in Mali Bamako, Koulikoro, Mali Collapse << |
| NCT00666679 | Asthma | Phase 2 | Completed | - | - |
| NCT00666679 | - | - | Completed | - | - |
| NCT03449836 | Allergy | Phase 3 | Not yet recruiting | December 31, 2018 | - |
| NCT03011632 | Sinusitis Ast... more >>hma Collapse << | Phase 4 | Recruiting | March 2019 | Poland ... more >> Department of Internal Medicine, Asthma and Allergy, Barlicki University Hospital, Medical University of Lodz, Lodz, Poland Recruiting Lodz, Poland, 90-153 Contact: Pawel Majak, MD, PhD Collapse << |
| NCT01401465 | Perennial Allergic Rhinitis | Phase 3 | Completed | - | United States, California ... more >> Asthma and Allergy Specialists Medical Group Huntington Beach, California, United States, 92647 Southern California Research Mission Veijo, California, United States, 92691 Allergy Associates Medical Group, Inc. San Diego, California, United States, 92120 Allergy & Asthma Medical Group & Research Center, A.P.C. San Diego, California, United States, 92123 United States, Colorado Asthma & Allergy Associates, P.C. Colorado Springs, Colorado, United States, 80907 United States, Massachusetts Northeast Medical Research Associates, Inc. North Dartmouth, Massachusetts, United States, 02747 United States, Minnesota Clinical Research Institute Inc. Minneappolis, Minnesota, United States, 55402 United States, Missouri The Clinical Research Center, L.L.C. St. Louis, Missouri, United States, 63141 United States, Nebraska The Asthma and Allergy Center, PC Bellevue, Nebraska, United States, 68123 United States, New Jersey Princeton Center for Clinical Research Skillman, New Jersey, United States, 08558 United States, Oregon Clinical Research Institute of Southern Oregon, PC Medford, Oregon, United States, 97504 Allergy Associates Research Center Portland, Oregon, United States, 97202 United States, Texas Pharmacuetical Research & Consulting, Inc. Dallas, Texas, United States, 75231 Western Sky Medical Research El Paso, Texas, United States, 79903 Sylvania Research San Antonio, Texas, United States, 78229 Collapse << |
| NCT01401465 | - | - | Completed | - | - |
| NCT03323866 | Chronic Rhinosinusitis (Diagno... more >>sis), Nasal Polyposis Collapse << | Phase 3 | Recruiting | October 2018 | Canada, Quebec ... more >> Université de Sherbrooke Recruiting Sherbrooke, Quebec, Canada, J1H 5N4 Contact: Marie Bussieres, MD FRCSC 819-346-1110 ext 14901 marie.bussieres@usherbrooke.ca Collapse << |
| NCT02478398 | Rhinitis, Allergic, Seasonal | Phase 3 | Completed | - | - |
| NCT00785278 | - | - | Unknown | July 2010 | United States, New Jersey ... more >> Kessler Medical Rehabilitation Research and Education Center Recruiting West Orange, New Jersey, United States, 07052 Contact: Sarah R. Dubowsky, Ph.D. 973-243-6838 sdubowsky@kmrrec.org Contact: Andrew M. Kwarciak, M.S. 973-243-6903 akwarciak@kmrrec.org Principal Investigator: Sarah R. Dubowsky, Ph.D. Sub-Investigator: Gail F. Forrest, Ph.D. Sub-Investigator: Trevor A. Dyson-Hudson, M.D. Sub-Investigator: Andrew M. Kwarciak, M.S. Sub-Investigator: Mathew B. Yarossi, B.S. Sub-Investigator: Arvind Ramanujam, M.S. Collapse << |
| 靶点 | Description | IC50 |
|---|

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
