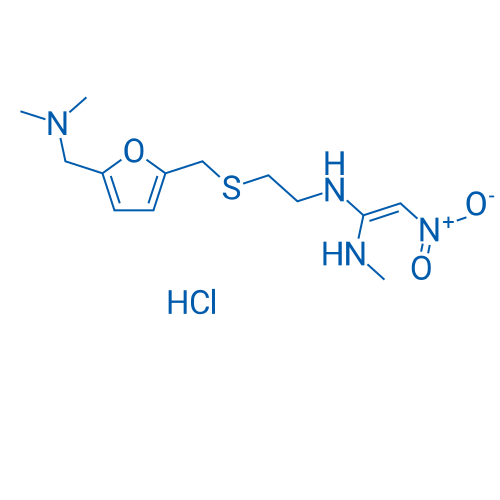
CAS No.: 66357-59-3
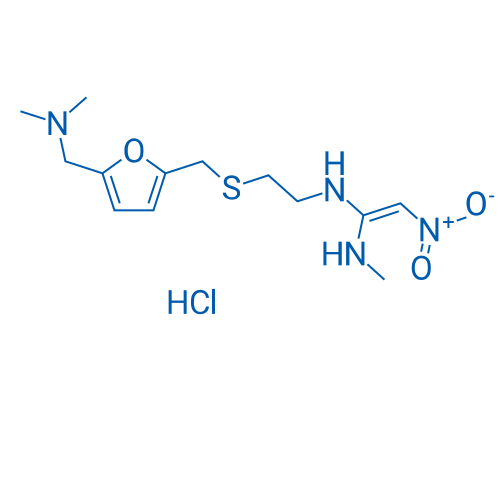
盐酸雷尼替丁 Catalog No. CSN13516
Synonyms: AH19065;Noctone;Ranitidine
Ranitidine HCl is an antagonist of histamine H2-receptor that can inhibit stomach acid production.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02655458 Multiple Myeloma Phase 1 Completed - United States, New York ... more >> Icahn School of Medicine at Mount Sinai New York, New York, United States, 10029 Collapse << NCT02125604 Relapsing-Remitting Multiple S... more >>clerosis Collapse << Phase 4 Completed - Germany ... more >> Research Site Augsburg, Germany Research Site Bamburg, Germany Research Site Bayreuth, Germany Research Site Berlin, Germany Research Site Bochum, Germany Research Site Bonn, Germany Research Site Erbach, Germany Research Site Erlangen, Germany Research Site Freiburg, Germany Research Site Hamburg, Germany Research Site Leipzig, Germany Research Site Marburg, Germany Research Site Minden, Germany Research Site Mittweida, Germany Research Site Munchen, Germany Research Site Munster, Germany Research Site Osnabruck, Germany Research Site Potsdam, Germany Research Site Siegen, Germany Collapse << NCT01017198 Advanced Solid Tumors Phase 1 Completed - United States, Arizona ... more >> Reseach Facility Scottsdale, Arizona, United States United States, California Reseach Facility Encinitas, California, United States United States, Texas Reseach Facility San Antonio, Texas, United States Collapse << - 更多
- 参考文献
- [1] Luyendyk JP, Maddox JF, et al. Ranitidine treatment during a modest inflammatory response precipitates idiosyncrasy-like liver injury in rats. J Pharmacol Exp Ther. 2003 Oct;307(1):9-16.
- [2] Herling AW, Becht M, et al. Inhibition of 14C-aminopyrine accumulation in isolated rabbit gastric glands by the H2-receptor antagonist HOE 760 (TZU-0460). Agents Actions. 1987 Feb;20(1-2):35-9.
- [3] Abduljabbar HN, Badr-Eldin SM, Aldawsari HM. Gastroretentive Ranitidine Hydrochloride Tablets with Combined Floating and Bioadhesive Properties: Factorial Design Analysis, In Vitro Evaluation and In Vivo Abdominal X-Ray Imaging. Curr Drug Deliv. 2015;12(5):578-90
- [4] Gaginella TS, Bauman JH. Ranitidine hydrochloride. Drug Intell Clin Pharm. 1983 Dec;17(12):873-85
- [5] Kamada S, Sakanoue M, Takeuchi M, Shimpo K, Tanabe T. [Effects of intravenous administration of ranitidine hydrochloride to the pregnant rat in organogenesis period]. J Toxicol Sci. 1984 Jun;9 Suppl 1:29-52. Japanese
- [6] Tamura J, Sato N, Ezaki H, Yokoyama S. [Teratological study on ranitidine hydrochloride in rabbits]. J Toxicol Sci. 1983 Jan;8 Suppl 1:141-50. Japanese
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 66357-59-3 | 储存条件 |
|
|||||
| 分子式 | C13H23ClN4O3S | 运输 | 蓝冰 | |||||
| 分子量 | 350.86 | 别名 | AH19065;Noctone;Ranitidine;Ranitidine (hydrochloride);Ranitidine Hydrochloride;ZANTAC;Tanidina;Toriol;Fendibina;Gastridina;Sostril;Zantic;Ranisen;Ranitidine HCl;盐酸雷尼替丁 | |||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02655458 | Multiple Myeloma | Phase 1 | Completed | - | United States, New York ... more >> Icahn School of Medicine at Mount Sinai New York, New York, United States, 10029 Collapse << |
| NCT02125604 | Relapsing-Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | Germany ... more >> Research Site Augsburg, Germany Research Site Bamburg, Germany Research Site Bayreuth, Germany Research Site Berlin, Germany Research Site Bochum, Germany Research Site Bonn, Germany Research Site Erbach, Germany Research Site Erlangen, Germany Research Site Freiburg, Germany Research Site Hamburg, Germany Research Site Leipzig, Germany Research Site Marburg, Germany Research Site Minden, Germany Research Site Mittweida, Germany Research Site Munchen, Germany Research Site Munster, Germany Research Site Osnabruck, Germany Research Site Potsdam, Germany Research Site Siegen, Germany Collapse << |
| NCT01017198 | Advanced Solid Tumors | Phase 1 | Completed | - | United States, Arizona ... more >> Reseach Facility Scottsdale, Arizona, United States United States, California Reseach Facility Encinitas, California, United States United States, Texas Reseach Facility San Antonio, Texas, United States Collapse << |
| NCT02125604 | - | - | Completed | - | - |
| NCT03319667 | Plasma Cell Myeloma | Phase 3 | Recruiting | January 2025 | - |
| NCT03368664 | Multiple Sclerosis | Phase 3 | Recruiting | March 2025 | - |
| NCT02205489 | Relapsing-remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | Belgium ... more >> Investigational Site Number 056002 Brugge, Belgium, 8000 Investigational Site Number 056001 Brussels, Belgium, 1070 France Investigational Site Number 250009 Dijon, France, 21000 Investigational Site Number 250005 Lille Cedex, France, 59037 Investigational Site Number 250001 Lyon Bron, France, 69677 Investigational Site Number 250006 Nancy, France Investigational Site Number 250010 Nantes, France, 44093 Investigational Site Number 250007 Nimes, France, 30029 Investigational Site Number 250008 PARIS Cedex 20, France, 75970 Investigational Site Number 250004 RENNES Cedex 9, France, 35033 Investigational Site Number 250003 Strasbourg Cedex 2, France, 67098 Investigational Site Number 250002 Toulouse, France, 31200 Netherlands Investigational Site Number 528001 Breda, Netherlands, 4818CK Investigational Site Number 528002 Rotterdam, Netherlands, 3079DZ Spain Investigational Site Number 724001 Barcelona, Spain, 08035 Investigational Site Number 724005 Bilbao, Spain, 48013 Investigational Site Number 724002 Madrid, Spain, 28040 Investigational Site Number 724006 Málaga, Spain, 29010 Investigational Site Number 724003 Sevilla, Spain, 41071 Investigational Site Number 724004 Valencia, Spain, 46009 Collapse << |
| NCT00004547 | Abdominal Neoplasm ... more >> Colonic Neoplasm Mesothelioma Peritoneal Neoplasm Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00004547 | - | - | Completed | - | - |
| NCT01011023 | Unnecessary Nasogastric Tube ... more >> Bowel Anastomosis Collapse << | Not Applicable | Completed | - | Mexico ... more >> Hospital Infantil de Mexico Mexico, DF, Mexico, 06720 Collapse << |
| NCT03518463 | Pregnancy Related | Not Applicable | Completed | - | Uganda ... more >> Mbarara university of Science and Technology Mbarara, Uganda Collapse << |
| NCT00655850 | Non-Small Cell Lung Cancer | Phase 2 | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35294 - 0104 Collapse << |
| NCT03541408 | Delirium | Not Applicable | Recruiting | March 1, 2019 | United States, Illinois ... more >> Rush University Medical Center Recruiting Chicago, Illinois, United States, 60612 Contact: David H Kim, MD 702-588-3470 david_h_kim@rush.edu Contact: Robert J McCarthy, MBA 312-942-7608 robert_j_mccarthy@rush.edu Collapse << |
| NCT02878512 | Success Rate Under Segmental E... more >>pidural Post Operative Pain Collapse << | Phase 4 | Completed | - | - |
| NCT02953210 | Pain, Postoperative ... more >> Anesthesia Complication Nausea Vomiting Ileus Paralytic Hemodynamic Instability Collapse << | Phase 4 | Unknown | March 2017 | Brazil ... more >> Faculdade de Ciências Médicas da Santa Casa de São Paulo Sao Paulo, Brazil, 01221-020 Faculdade de Ciências Médicas da Santa Casa de São Paulo Sao Paulo, Brazil, 01333000 Collapse << |
| NCT00655850 | - | - | Completed | - | - |
| NCT01710592 | Gastro Oesophageal Cancer | Phase 2 | Completed | - | Ireland ... more >> Mercy University Hospital Cork, Ireland Beaumont Hospital Dublin, Ireland Mater Misericordiae University hospital & Mater Private Hospital Dublin, Ireland St James's Hospital Dublin, Ireland The Adelaide and Meath Hospital Dublin, Ireland Waterford Regional Hospital Waterford, Ireland Collapse << |
| NCT03591120 | Delirium | Not Applicable | Recruiting | March 1, 2019 | United States, Illinois ... more >> Rush University Medical Center Recruiting Chicago, Illinois, United States, 60612 Contact: Robert J McCarthy, PharmD 312-942-7608 Robert_J_McCarthy@rush.edu Contact: David H Kim, MD 702-588-3470 david_h_kim@rush.edu Collapse << |
| NCT00924911 | Infections, Bacterial | Phase 1 | Completed | - | United States, New York ... more >> GSK Investigational Site Buffalo, New York, United States, 14202 Collapse << |
| NCT03710304 | Cesarean Section Complications | Not Applicable | Recruiting | June 1, 2020 | Egypt ... more >> Aswan University Recruiting Aswan, Egypt, 81528 Contact: hany f sallam, md 01092440504 ext 002 nahla.elsayed@aswu.ed.eg Collapse << |
| NCT02541643 | - | - | - | - | United States, New York ... more >> Icahn School.of Medicine at Mount Sinai New York, New York, United States, 10029 Collapse << |
| NCT01411410 | Neoplasms | Phase 1 | Completed | - | United States, Missouri ... more >> Saint Louis, Missouri, United States, 63110 United States, New York New York, New York, United States, 10065 United States, Texas Houston, Texas, United States, 77030 Collapse << |
| NCT02360696 | - | - | Unknown | July 2016 | United States, Missouri ... more >> Children's Mercy Recruiting Kansas City, Missouri, United States, 64108 Contact: Nancy E. Lathrom, CCRC 816-855-1860 nelathrom@cmh.edu Contact: Craig A. Friesen, MD 816.234.3016 cfriesen@cmh.edu Sub-Investigator: Jennifer V. Schurman, PhD Principal Investigator: Steven Leeder, PharmD, PhD Principal Investigator: Craig A. Friesen, MD Sub-Investigator: Shui Q. Ye, MD, PhD Sub-Investigator: Nancy E. Lathrom, CCRC Sub-Investigator: Trevor J. Cole, BS Collapse << |
| NCT02349763 | Hypersensitivity Reaction | Phase 3 | Unknown | August 2015 | Thailand ... more >> Rajavithi Hospital Bangkok, Thailand, 10400 Collapse << |
| NCT01074437 | Hemangioma | Phase 2 | Terminated(Insufficient enroll... more >>ment) Collapse << | - | United States, Washington ... more >> Seattle Children's Hospital Seattle, Washington, United States, 98105 Collapse << |
| NCT01074437 | - | - | Terminated(Insufficient enroll... more >>ment) Collapse << | - | - |
| NCT01734759 | Healthy | Phase 1 | Completed | - | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT03301454 | Esophageal Cancer, Squamous Ce... more >>ll Collapse << | Phase 2 | Recruiting | February 2022 | France ... more >> Centre Hospitalier Universitaire Not yet recruiting Amiens, France, 80080 Contact: Vincent HAUTEFEUILLE, MD +33322088849 heutefeuille.vincent@chu-amiens.fr Principal Investigator: Vincent HAUTEFEUILLE, MD Centre Paul Papin Not yet recruiting Angers, France, 49055 Contact: Olivier CAPITAIN, MD +33240679900 olivier.capitain@ico.unicancer.fr Principal Investigator: Olivier CAPITAIN, MD Sub-Investigator: Véronique GUERIN-MEYER, MD Sub-Investigator: Julie VANBOCKSTAEL, MD Centre François Baclesse Not yet recruiting Caen, France, 14076 Contact: Marie-Pierre GALAIS, MD +33231455016 mp.galais@baclesse.fr Principal Investigator: Marie-Pierre GALAIS, MD Sub-Investigator: Françoise POLYCARPE-OSAER, MD Sub-Investigator: Aurélie PARZY, MD Sub-Investigator: Stéphane CORBINAIS, MD Centre Oscar Lambret Recruiting Lille, France, 59020 Contact: Farid EL HAJBI, MD +33320295266 f-elhajbi@o-lambret.fr Sub-Investigator: Frederik LAESTADIUS, MD Sub-Investigator: Diane PANNIER, MD Sub-Investigator: Natacha STERN, MD Sub-Investigator: Nicolas PENEL, PhD Sub-Investigator: Thomas RYCKEWAERT, MD Centre René Gauducheau Not yet recruiting Nantes, France, 44805 Contact: Olivier CAPITAIN, MD +33240679900 olivier.capitain@ico.unicancer.fr Sub-Investigator: Hélène SENELLART, MD Sub-Investigator: Sandrine HIRET, MD Principal Investigator: Olivier CAPITAIN, MD Sub-Investigator: Judith RAIMBOURG, MD Centre Armoricain de Radiothérapie, Imagerie médicale et Oncologie Not yet recruiting Plérin, France, 22190 Contact: Pierre-Luc ETIENNE, MD +33296752216 pl.etienne@cario-sante.fr Principal Investigator: Pierre-Luc ETIENNE, MD Sub-Investigator: Jérôme MARTIN-BABAU, MD Sub-Investigator: Dominique BESSON, MD Sub-Investigator: Anne-Claire HARDY-BESSARD, MD Centre Eugène Marquis Not yet recruiting Rennes, France, 35042 Contact: Julien EDELINE, Pr +33299253000 j.edeline@rennes.unicancer.fr Principal Investigator: Samuel LE SOURD, MD Sub-Investigator: Anais BODERE, MD Sub-Investigator: Julien EDELINE, PhD Sub-Investigator: Claire LARIBLE, MD Sub-Investigator: Fanny LE DU, MD Sub-Investigator: Astrid LIEVRE, MD Sub-Investigator: Marc PRACHT, MD Collapse << |
| NCT01048229 | Early-stage Parkinson's Diseas... more >>e Collapse << | Phase 4 | Terminated(recruitment) | - | - |
| NCT00551122 | Brain and Central Nervous Syst... more >>em Tumors Extragonadal Germ Cell Tumor Ovarian Cancer Testicular Germ Cell Tumor Collapse << | Phase 1 Phase 2 | Unknown | - | United Kingdom ... more >> Queen Elizabeth Hospital at University Hospital of Birmingham NHS Trust Recruiting Birmingham, England, United Kingdom, B15 2TH Contact: Michael H. Cullen, MD 0121-627-2444 Southampton General Hospital Recruiting Southampton, England, United Kingdom, SO16 6YD Contact: G. Mead, MD 44-23-8079-8639 Royal Marsden - Surrey Recruiting Sutton, England, United Kingdom, SM2 5PT Contact: Robert A. Huddart, MD 44-20-8661-3457 robert.huddart@icr.ac.uk Collapse << |
| NCT00855660 | Pharmacology, Clinical | Phase 1 | Completed | - | Germany ... more >> Köln, Nordrhein-Westfalen, Germany, 51147 Collapse << |
| NCT01032655 | Helicobacter Pylori Infection | Phase 4 | Completed | - | Taiwan ... more >> National Taiwan University Hospital Taipei, Taiwan, 10002 Collapse << |
| NCT01248962 | Ovarian Cancer ... more >> Fallopian Tube Cancer Peritoneal Cancer Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT00885417 | H. Pylori Infection | Phase 4 | Completed | - | Taiwan ... more >> National Taiwan University Hospital Taipei, Taiwan, 10002 Collapse << |
| NCT00001910 | - | - | Completed | - | United States, Maryland ... more >> National Institute of Allergy and Infectious Diseases (NIAID) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT03241602 | Decrease Post Laproscopy Shoul... more >>der and Abdominal Pain Collapse << | Phase 4 | Completed | - | Egypt ... more >> Faculty of medicine CAIRO UNIVERISTY Cairo, Egypt Collapse << |
| NCT00443963 | Dyspepsia | Phase 4 | Terminated(The Principal Inves... more >>tigator is no longer affiliated with the Study Site.) Collapse << | - | United States, District of Col... more >>umbia Washington Hospital Center 110 Irving St. NW, Washington, District of Columbia, United States, 20010 Collapse << |
| NCT00985608 | Helicobacter Pylori Infection | Phase 2 | Unknown | September 2009 | Italy ... more >> Institute of Internal Medicine - Catholic University Recruiting Rome, Italy, 00168 Contact: Giovanni Cammarota, MD 0039-06-30155948 gcammarota@rm.unicatt.it Principal Investigator: Giovanni Cammarota, MD Collapse << |
| NCT03644550 | Mesothelioma | Phase 2 | Recruiting | February 28, 2023 | United States, Maryland ... more >> National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT00285688 | - | - | Unknown | December 2008 | Thailand ... more >> Rajavithi Hospital Recruiting Ratchathewi, Bangkok, Thailand, 10400 Contact: Piyathida Harnsomboonrana, M.D. 6618423854 pboontee@yahoo.com Collapse << |
| NCT02570529 | Pancreatic Ductal Adenocarcino... more >>ma Collapse << | Phase 4 | Unknown | February 2018 | - |
| NCT01866449 | Glioblastoma Multiforme (GBM) ... more >>WHO Grade IV Collapse << | Phase 2 | Completed | - | Germany ... more >> Hämatologisch onkologische Praxis Augsburg, Germany, 86150 Stiftungsklinikum Mittelrhein GmbH Koblenz, Germany, 56068 Lars Bullinger, MD Ulm, Germany, 89081 Collapse << |
| NCT01028807 | Early Feeding ... more >> Bowel Anastomosis Collapse << | Not Applicable | Completed | - | Mexico ... more >> Hospital Infantil de Mexico Mexico Df, DF, Mexico, 06720 Collapse << |
| NCT01083537 | Ovarian Cancer ... more >> Peritoneal Cancer Fallopian Tube Cancer Bowel Obstruction Collapse << | Phase 1 Phase 2 | Terminated(Slow accrual) | - | Canada, Ontario ... more >> Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT03715881 | Non-Arteritic Anterior Ischemi... more >>c Optic Neuropathy (NAION) Collapse << | Phase 2 | Recruiting | March 1, 2019 | Iran, Islamic Republic of ... more >> Ophthalmic Research Center Recruiting Tehran, Iran, Islamic Republic of Contact: Homayon Nikkhah, MD 009822591616 labbafi@hotmail.com Collapse << |
| NCT00037570 | Peptic Ulcer Hemorrhage | Phase 2 | Completed | - | United States, California ... more >> Los Angeles, California, United States, 90073 San Diego, California, United States, 62103-8401 United States, Ohio Cincinnati, Ohio, United States, 45267-0595 United States, Pennsylvania Philadelphia, Pennsylvania, United States, 19140 United States, Texas Houston, Texas, United States, 77030-4211 United States, Virginia Norfolk, Virginia, United States, 23507 Collapse << |
| NCT02441894 | Prostate Cancer | Phase 4 | Completed | - | Japan ... more >> Investigational Site Number 392004 Chuo-ku, Chiba, Japan Investigational Site Number 392008 Kita-gun, Japan Investigational Site Number 392007 Kobe-shi, Hyogo, Japan Investigational Site Number 392005 Nagakute-shi, Aichi, Japan Investigational Site Number 392006 Osaka Sayama-shi, Osaka, Japan Investigational Site Number 392001 Shinjuku-ku, Tokyo, Japan Investigational Site Number 392002 Yokohama-shi, Kanagawa, Japan Investigational Site Number 392009 Yokohama-shi, Japan Collapse << |
| NCT00199342 | Stage IV Melanoma | Phase 1 Phase 2 | Terminated(Occurrence of 2 dos... more >>e-limiting toxicities at the initial dose level (60 mg/m2) in Part I.) Collapse << | - | United States, Alabama ... more >> Comprehensive Cancer Center- University of Alabama at Birmingham Birmingham, Alabama, United States, 35294 United States, Florida H. Lee Moffitt Cancer Center Tampa, Florida, United States, 22612 Collapse << |
| NCT02555852 | - | - | Completed | - | Canada, Quebec ... more >> Lady Davis Institute for Medical Research, Jewish General Hospital Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT00247715 | Dyspepsia Gas... more >>trointestinal Diseases Collapse << | Not Applicable | Completed | - | Netherlands ... more >> Radboud University Nijmegen Medical Center Nijmegen, Gelderland, Netherlands, 6500 HB Maastricht University Maastricht, Limburg, Netherlands, 6200 MB UMC Utrecht Utrecht, Netherlands Collapse << |
| NCT02169856 | Postoperative Nausea and Vomit... more >>ing Collapse << | Not Applicable | Completed | - | Pakistan ... more >> Benazir Bhutto Hospital Rawalpindi. Rawalpindi, Punjab, Pakistan, 46000 Collapse << |
| NCT00930670 | Coronary Artery Disease | Phase 4 | Completed | - | Canada, Quebec ... more >> Institut Universitaire de Cardiologie et de Pneumologie de Quebec Quebec City, Quebec, Canada, G1V4G5 Collapse << |
| NCT03619408 | - | - | Not yet recruiting | July 2020 | - |
| NCT02441673 | Post Anaesthetic Shivering | Phase 2 | Not yet recruiting | June 2019 | - |
| NCT01209455 | Poisoning | Not Applicable | Unknown | September 2012 | United Kingdom ... more >> Clinical Research Facility, Royal Infirmary of Edinburgh Recruiting Edinburgh, Midlothian, United Kingdom, EH16 4SA Principal Investigator: Euan A Sandilands, MRCP BSc Collapse << |
| NCT00220909 | Patients Undergoing Elective C... more >>oronary Artery Bypass Graft Collapse << | Phase 4 | Terminated(Recruitment slow) | - | United States, Pennsylvania ... more >> Temple University School of Medicine Philadelphia, Pennsylvania, United States, 19140 Collapse << |
| NCT02999633 | T-cell Type Acute Leukemia-Pre... more >>cursor T-lymphoblastic Lymphoma/Leukaemia Collapse << | Phase 2 | Terminated(Due to an unsatisfa... more >>ctory benefit/risk ratio, as specified in & 14.8.1 of the protocol, Sanofi decided to stop enrollment and terminate ACT14596 prematurely) Collapse << | - | United States, Georgia ... more >> Investigational Site Number 8400002 Atlanta, Georgia, United States, 30342 United States, New Jersey Investigational Site Number 8400003 Hackensack, New Jersey, United States, 07601 United States, Texas Investigational Site Number 8400001 Houston, Texas, United States, 77030 Finland Investigational Site Number 2460001 Helsinki, Finland, 00029 France Investigational Site Number 2500005 Nantes Cedex 01, France, 44093 Investigational Site Number 2500001 Paris Cedex 10, France, 75475 Investigational Site Number 2500004 Pessac, France, 33600 Investigational Site Number 2500002 Pierre Benite, France, 69310 Hungary Investigational Site Number 3480001 Budapest, Hungary, 1083 Investigational Site Number 3480003 Budapest, Hungary, 1097 Investigational Site Number 3480002 Debrecen, Hungary, 4032 Italy Investigational Site Number 3800001 Bergamo, Italy, 24127 Investigational Site Number 3800004 Brescia, Italy, 25123 Lithuania Investigational Site Number 4400001 Vilnius, Lithuania, 08661 Russian Federation Investigational Site Number 6430003 Moscow, Russian Federation, 117198 Investigational Site Number 6430004 Moscow, Russian Federation, 125167 Investigational Site Number 6430001 Moscow, Russian Federation, 129301 Collapse << |
| NCT01274130 | Healthy Adult | Not Applicable | Completed | - | Korea, Republic of ... more >> Severance Hospital Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT02966665 | Chronic Obstructive Pulmonary ... more >>Disease Pulmonary Artery Hypertension Heart Failure Hypertension Collapse << | Phase 1 | Recruiting | August 2020 | United States, Utah ... more >> George E Wahlen VA Medical Center Recruiting Salt Lake City, Utah, United States, 84132 Contact: Russell Richardson, Ph.D. 801-582-1565 r.richardson@hsc.utah.edu Contact: Ashley Nelson, MD 801-582-1565 ext 4127 ash.nelson@hsc.utah.edu Collapse << |
| NCT02123498 | Eustachian Tube Dysfunction ... more >> Laryngopharyngeal Reflux Collapse << | Phase 4 | Withdrawn(IRB not approved) | - | United States, California ... more >> 11234 Anderson Street Loma Linda, California, United States, 92354 1895 Orange Tree Lane, Suite 102 Redlands, California, United States, 92374 Collapse << |
| NCT00223691 | Autonomic Failure ... more >> Orthostatic Hypotension Collapse << | Phase 1 | Completed | - | United States, Tennessee ... more >> Vanderbilt University Nashville, Tennessee, United States, 37232 Collapse << |
| NCT02004197 | Helicobacter Pylori [H. Pylori... more >>] as the Cause of Diseases Collapse << | Phase 2 Phase 3 | Completed | - | Pakistan ... more >> clinical trial was conducted in high risk areas of Pakistan such as Karachi, Bahawalpur, Islamabad and Rawalpindi at Shifa-Ul-Mulk Memorial Hospital, Hamdrad University Karachi, Bahawalpur Victoria Hospital and Nawaz Salik Hospital respectively Karachi, Pakistan Shifa Ul MUlk Memorial Hospital Karachi, Pakistan Collapse << |
| NCT00131248 | Gastroesophageal Reflux | Phase 3 | Completed | - | United States, Texas ... more >> Memorial Hermann Children's Hospital Houston, Texas, United States, 77030 Collapse << |
| NCT01261546 | Parapneumonic Pleural Effusion... more >> Empyema Collapse << | Phase 2 | Completed | - | Spain ... more >> Hospital Carlos Haya Malaga, Andalucia, Spain Complejo Hospitalario Toledo Toledo, Castilla La Mancha, Spain Hospital Principe de Asturias Alcalá de Henares, Madrid, Spain Hospital Universitario de Getafe Getafe, Madrid, Spain Hospital Infanta Sofia San Sebastián de los Reyes, Madrid, Spain, 28014 Hospital Infantil La Paz Madrid, Spain, 28037 Hospital Ramón y Cajal Madrid, Spain Hospital Universitario Gregorio Marañón Madrid, Spain Collapse << |
| NCT02258815 | Neuroblastoma Recurrent | Phase 2 | Unknown | December 2015 | Austria ... more >> University Hospital Graz Recruiting Graz, Austria, 8036 Contact: Wolfgang Schwinger, MD, PhD +43 31638512605 wolfgang.schwinger@medunigraz.at Principal Investigator: Wolfgang Schwinger, MD, PhD St. Anna Childrens Hospital Recruiting Vienna, Austria, 1090 Contact: Ruth Ladenstein, MD, PhD 43 1 40470 3250 ruth.ladenstein@ccri.at Principal Investigator: Ruth Ladenstein, MD, PhD Germany University Hospital Greifswald Recruiting Greifswald, Germany, 17475 Contact: Holger Lode, MD, PhD +49 3834866301 lode@uni-greifswald.de Principal Investigator: Holger Lode, MD, PhD University Hospital Tuebingen Recruiting Tuebingen, Germany, 72076 Contact: Peter Lang, MD, PhD +4970712983781 peter.lang@med.uni-tuebingen.de Principal Investigator: Peter Lang, MD, PhD Collapse << |
| NCT00161200 | Esophageal Metaplasia | Phase 3 | Terminated(slow accrual) | - | United States, New Jersey ... more >> Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08901 Collapse << |
| NCT01332630 | Breast Cancer | Phase 2 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01332630 | - | - | Completed | - | - |
| NCT00131248 | - | - | Completed | - | - |
| NCT02703896 | Pulmonary Aspiration of Gastri... more >>c Contents Collapse << | Phase 4 | Completed | - | - |
| NCT00030992 | - | - | Completed | - | - |
| NCT00030992 | Renal Cell Carcinoma | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT03667703 | Congenital Heart Disease ... more >> Upper Gastrointestinal Bleeding Stress Ulcer Infection Collapse << | Not Applicable | Not yet recruiting | June 30, 2021 | United States, Massachusetts ... more >> Boston Children's Hospital Not yet recruiting Boston, Massachusetts, United States, 02115 Contact: Kimberly I Mills, MD 617-355-7866 kimberly.mills@cardio.chboston.org Contact: Ben D Albert, MD 617-355-7866 ben.albert@childrens.harvard.edu Collapse << |
| NCT03583567 | Protamine Adverse Reaction | Phase 4 | Recruiting | December 1, 2020 | Thailand ... more >> Siriraj Hospital Recruiting Bangkok, Thailand, 10700 Contact: Sirilak Suksompoong, MD ssuksompong5@gmail.com Collapse << |
| NCT03468777 | Healthy | Phase 1 | Terminated(The study has been ... more >>terminated due to pending data analysis) Collapse << | - | Belgium ... more >> Clinical Pharmacology Unit Merksem, Belgium, 2170 Collapse << |
| NCT00585351 | - | - | Completed | - | - |
| NCT00585351 | Stroke | Not Applicable | Completed | - | United States, Iowa ... more >> University of Iowa Iowa City, Iowa, United States, 52242 Collapse << |
| NCT00590928 | Critically Ill Patients ... more >> Indication for Stress Ulcer Prophylaxis Collapse << | Phase 4 | Completed | - | Austria ... more >> Medical University of Vienna, Department of Medicine III, ICU Vienna, Austria, 1090 Collapse << |
| NCT00000964 | HIV Infections | Phase 1 | Completed | - | United States, Maryland ... more >> Johns Hopkins Adult AIDS CRS Baltimore, Maryland, United States, 21205 Collapse << |
| NCT00405119 | Gastroesophageal Reflux Diseas... more >>e Reflux, Gastroesophageal Collapse << | Phase 2 | Completed | - | Australia, New South Wales ... more >> GSK Investigational Site Randwick, New South Wales, Australia, 2031 Australia, Queensland GSK Investigational Site Herston, Queensland, Australia, 4006 Collapse << |
| NCT03140033 | Hemorrhage Postpartum | Phase 2 | Recruiting | May 2017 | Egypt ... more >> Ain shams maternity hospital Recruiting Cairo, Egypt Contact: Mohamed Fathy, M.B.B.Ch 00201099984100 Mohamedfathy31085@gmail.com Contact: AMR Helmy, MD.MRCOG 00201227900014 am_helmy77@hotmail.com Collapse << |
| NCT03011463 | Pharmacokinetics ... more >> Inhibition Enzyme Drug Interaction Potentiation Collapse << | Phase 1 | Completed | - | - |
| NCT00702871 | Ventilator Associated Pneumoni... more >>a Etiological Organisms Antimicrobial Drug Susceptibility Pattern Stress Ulcer Prophylaxis Collapse << | Phase 4 | Completed | - | India ... more >> Maulana Azad Medical College and Lok Nayak Hospital Delhi, India, 110002 Collapse << |
| NCT01976078 | - | - | Completed | - | United States, California ... more >> Children's Hospital Los Angeles Los Angeles, California, United States, 90027 Collapse << |
| NCT00668317 | - | - | Terminated(Primary care physic... more >>ians began prescribing antacid therapy for chronic cough) Collapse << | - | - |
| NCT00668317 | Cough | Phase 3 | Terminated(Primary care physic... more >>ians began prescribing antacid therapy for chronic cough) Collapse << | - | United Kingdom ... more >> Clinical Trials Unit , Cardiovascular and respiratory studies, Castle Hill Hospital Cottingham, East Yorkshire, United Kingdom, HU16 5JQ Collapse << |
| NCT02172417 | Healthy | Phase 1 | Completed | - | - |
| NCT00633412 | NSAID Associated Gastric Ulcer... more >>s Collapse << | Phase 3 | Completed | - | - |
| NCT00839306 | Gastroesophageal Reflux Diseas... more >>e (GERD) Collapse << | Phase 3 | Completed | - | United States, Illinois ... more >> Associates, Ltd. Moline, Illinois, United States, 61265 Midwest Clinical Moline, Illinois, United States, 61265 Research Moline, Illinois, United States, 61265 Moline, Illinois, United States, 61265 Collapse << |
| NCT02733640 | Antiplatelet Effect | Not Applicable | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seongnam, Gyeonggi-do, Korea, Republic of, 463-707 Collapse << |
| NCT00838526 | Gastroesophageal Reflux Diseas... more >>e (GERD) Collapse << | Phase 3 | Completed | - | United States, Illinois ... more >> Ltd. Moline, Illinois, United States, 61265 Midwest Clinical Moline, Illinois, United States, 61265 Research Associates Moline, Illinois, United States, 61265 Moline, Illinois, United States, 61265 Collapse << |
| NCT00839306 | - | - | Completed | - | - |
| NCT00838526 | - | - | Completed | - | - |
| NCT01908257 | Healthy | Phase 1 | Completed | - | United States, California ... more >> Cypress, California, United States, 90630 Collapse << |
| NCT01392755 | Healthy Volunteer | Phase 1 | Completed | - | United States, Kansas ... more >> Lenexa, Kansas, United States, 66219 Collapse << |
| NCT01538797 | Reflux Oesophagitis | Phase 1 | Completed | - | United Kingdom ... more >> Hammersmith Medicines Research London, United Kingdom, NW10 7EW Collapse << |
| NCT00401752 | Gastric Ulcer | Phase 3 | Completed | - | China, Fujian ... more >> Research Site Fuzhou, Fujian, China China, Guangdong Research Site Guangzhou, Guangdong, China China, Hubei Research Site Wuhan, Hubei, China China, Jiangsu Research Site Suzhou, Jiangsu, China China, Liaoning Research Site Shenyang, Liaoning, China China, Shanxi Research Site Xi'an, Shanxi, China China Research Site Beijing, China Research Site Shanghai, China Hong Kong Research Site Hong Kong, Hong Kong Collapse << |
| NCT00633672 | NSAID Associated Gastric Ulcer... more >>s Collapse << | Phase 3 | Completed | - | - |
| NCT03145012 | Cancer | Phase 4 | Not yet recruiting | April 30, 2019 | Canada, Nova Scotia ... more >> Nova Scotia Health Authority Not yet recruiting Halifax, Nova Scotia, Canada, B3H 1V7 Contact: Lisa Barrett, MD lisa.barrett@nshealth.ca Contact: Jill Moore jill.moore@nshealth.ca Collapse << |
| NCT02170792 | Healthy | Phase 1 | Completed | - | - |
| NCT01682408 | Pharmacokinetics | Phase 1 | Completed | - | United States, Kansas ... more >> Overland Park, Kansas, United States Collapse << |
| NCT00401752 | - | - | Completed | - | - |
| NCT01737840 | Dyspepsia | Phase 4 | Completed | - | Turkey ... more >> Akdeniz University Antalya, Turkey, 07050 Collapse << |
| NCT00002106 | HIV Infections | Phase 2 | Completed | - | United States, California ... more >> Pacific Oaks Med Group Sherman Oaks, California, United States, 91403 United States, Florida Community Research Initiative Coral Gables, Florida, United States, 33146 United States, North Carolina Duke Univ Med Ctr Durham, North Carolina, United States, 27710 United States, Texas Houston Clinical Research Network Houston, Texas, United States, 77006 Collapse << |
| NCT01539655 | Medullary Thyroid Cancer | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT00527878 | JOB's Syndrome ... more >> Hyper-IgE Recurrent Infection Syndrome Immune Deficiency Collapse << | Phase 2 | Terminated(Failure to enroll a... more >>dequate patient numbers due to small number of eligible patients) Collapse << | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00527878 | - | - | Terminated(Failure to enroll a... more >>dequate patient numbers due to small number of eligible patients) Collapse << | - | - |
| NCT01896557 | Coronary Artery Disease ... more >> Drug Interaction Potentiation Collapse << | Phase 4 | Completed | - | - |
| NCT02700087 | Laryngomalacia ... more >> Acid Reflux Stridor Collapse << | Not Applicable | Withdrawn | February 2017 | United States, California ... more >> Lucile Packard Children's Hospital Stanford Palo Alto, California, United States, 94304 Collapse << |
| NCT02197143 | Dyspepsia | Phase 4 | Completed | - | Turkey ... more >> Pamukkale Universty Denizli, Turkey, 20000 Collapse << |
| NCT00247130 | Peptic Ulcers | Phase 4 | Withdrawn(Superiority of iv om... more >>eprazole to ranitidine has already been proven by others.) Collapse << | - | Japan ... more >> Department of Internal Medicine, Keio University School of Medicine Tokyo, Japan, 1608582 Collapse << |
| NCT01131702 | Healthy | Phase 1 | Completed | - | Canada, Quebec ... more >> Anapharm Inc. Montreal, Quebec, Canada, G1V 2K8 Collapse << |
| NCT02195804 | Healthy | Phase 1 | Completed | - | - |
| NCT01737840 | - | - | Completed | - | - |
| NCT01896557 | - | - | Completed | - | - |
| NCT02655458 | Multiple Myeloma | Phase 1 | Completed | - | United States, New York ... more >> Icahn School of Medicine at Mount Sinai New York, New York, United States, 10029 Collapse << |
| NCT02125604 | Relapsing-Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | Germany ... more >> Research Site Augsburg, Germany Research Site Bamburg, Germany Research Site Bayreuth, Germany Research Site Berlin, Germany Research Site Bochum, Germany Research Site Bonn, Germany Research Site Erbach, Germany Research Site Erlangen, Germany Research Site Freiburg, Germany Research Site Hamburg, Germany Research Site Leipzig, Germany Research Site Marburg, Germany Research Site Minden, Germany Research Site Mittweida, Germany Research Site Munchen, Germany Research Site Munster, Germany Research Site Osnabruck, Germany Research Site Potsdam, Germany Research Site Siegen, Germany Collapse << |
| NCT01017198 | Advanced Solid Tumors | Phase 1 | Completed | - | United States, Arizona ... more >> Reseach Facility Scottsdale, Arizona, United States United States, California Reseach Facility Encinitas, California, United States United States, Texas Reseach Facility San Antonio, Texas, United States Collapse << |
| NCT00655850 | Non-Small Cell Lung Cancer | Phase 2 | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35294 - 0104 Collapse << |
| NCT03319667 | Plasma Cell Myeloma | Phase 3 | Recruiting | January 2025 | - |
| NCT03368664 | Multiple Sclerosis | Phase 3 | Recruiting | March 2025 | - |
| NCT02125604 | - | - | Completed | - | - |
| NCT00655850 | - | - | Completed | - | - |
| NCT02953210 | Pain, Postoperative ... more >> Anesthesia Complication Nausea Vomiting Ileus Paralytic Hemodynamic Instability Collapse << | Phase 4 | Unknown | March 2017 | Brazil ... more >> Faculdade de Ciências Médicas da Santa Casa de São Paulo Sao Paulo, Brazil, 01221-020 Faculdade de Ciências Médicas da Santa Casa de São Paulo Sao Paulo, Brazil, 01333000 Collapse << |
| NCT00004547 | Abdominal Neoplasm ... more >> Colonic Neoplasm Mesothelioma Peritoneal Neoplasm Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02205489 | Relapsing-remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | Belgium ... more >> Investigational Site Number 056002 Brugge, Belgium, 8000 Investigational Site Number 056001 Brussels, Belgium, 1070 France Investigational Site Number 250009 Dijon, France, 21000 Investigational Site Number 250005 Lille Cedex, France, 59037 Investigational Site Number 250001 Lyon Bron, France, 69677 Investigational Site Number 250006 Nancy, France Investigational Site Number 250010 Nantes, France, 44093 Investigational Site Number 250007 Nimes, France, 30029 Investigational Site Number 250008 PARIS Cedex 20, France, 75970 Investigational Site Number 250004 RENNES Cedex 9, France, 35033 Investigational Site Number 250003 Strasbourg Cedex 2, France, 67098 Investigational Site Number 250002 Toulouse, France, 31200 Netherlands Investigational Site Number 528001 Breda, Netherlands, 4818CK Investigational Site Number 528002 Rotterdam, Netherlands, 3079DZ Spain Investigational Site Number 724001 Barcelona, Spain, 08035 Investigational Site Number 724005 Bilbao, Spain, 48013 Investigational Site Number 724002 Madrid, Spain, 28040 Investigational Site Number 724006 Málaga, Spain, 29010 Investigational Site Number 724003 Sevilla, Spain, 41071 Investigational Site Number 724004 Valencia, Spain, 46009 Collapse << |
| NCT03518463 | Pregnancy Related | Not Applicable | Completed | - | Uganda ... more >> Mbarara university of Science and Technology Mbarara, Uganda Collapse << |
| NCT01011023 | Unnecessary Nasogastric Tube ... more >> Bowel Anastomosis Collapse << | Not Applicable | Completed | - | Mexico ... more >> Hospital Infantil de Mexico Mexico, DF, Mexico, 06720 Collapse << |
| NCT00004547 | - | - | Completed | - | - |
| NCT01411410 | Neoplasms | Phase 1 | Completed | - | United States, Missouri ... more >> Saint Louis, Missouri, United States, 63110 United States, New York New York, New York, United States, 10065 United States, Texas Houston, Texas, United States, 77030 Collapse << |
| NCT03591120 | Delirium | Not Applicable | Recruiting | March 1, 2019 | United States, Illinois ... more >> Rush University Medical Center Recruiting Chicago, Illinois, United States, 60612 Contact: Robert J McCarthy, PharmD 312-942-7608 Robert_J_McCarthy@rush.edu Contact: David H Kim, MD 702-588-3470 david_h_kim@rush.edu Collapse << |
| NCT02878512 | Success Rate Under Segmental E... more >>pidural Post Operative Pain Collapse << | Phase 4 | Completed | - | - |
| NCT02349763 | Hypersensitivity Reaction | Phase 3 | Unknown | August 2015 | Thailand ... more >> Rajavithi Hospital Bangkok, Thailand, 10400 Collapse << |
| NCT00924911 | Infections, Bacterial | Phase 1 | Completed | - | United States, New York ... more >> GSK Investigational Site Buffalo, New York, United States, 14202 Collapse << |
| NCT01710592 | Gastro Oesophageal Cancer | Phase 2 | Completed | - | Ireland ... more >> Mercy University Hospital Cork, Ireland Beaumont Hospital Dublin, Ireland Mater Misericordiae University hospital & Mater Private Hospital Dublin, Ireland St James's Hospital Dublin, Ireland The Adelaide and Meath Hospital Dublin, Ireland Waterford Regional Hospital Waterford, Ireland Collapse << |
| NCT02541643 | - | - | - | - | United States, New York ... more >> Icahn School.of Medicine at Mount Sinai New York, New York, United States, 10029 Collapse << |
| NCT02360696 | - | - | Unknown | July 2016 | United States, Missouri ... more >> Children's Mercy Recruiting Kansas City, Missouri, United States, 64108 Contact: Nancy E. Lathrom, CCRC 816-855-1860 nelathrom@cmh.edu Contact: Craig A. Friesen, MD 816.234.3016 cfriesen@cmh.edu Sub-Investigator: Jennifer V. Schurman, PhD Principal Investigator: Steven Leeder, PharmD, PhD Principal Investigator: Craig A. Friesen, MD Sub-Investigator: Shui Q. Ye, MD, PhD Sub-Investigator: Nancy E. Lathrom, CCRC Sub-Investigator: Trevor J. Cole, BS Collapse << |
| NCT03541408 | Delirium | Not Applicable | Recruiting | March 1, 2019 | United States, Illinois ... more >> Rush University Medical Center Recruiting Chicago, Illinois, United States, 60612 Contact: David H Kim, MD 702-588-3470 david_h_kim@rush.edu Contact: Robert J McCarthy, MBA 312-942-7608 robert_j_mccarthy@rush.edu Collapse << |
| NCT01074437 | Hemangioma | Phase 2 | Terminated(Insufficient enroll... more >>ment) Collapse << | - | United States, Washington ... more >> Seattle Children's Hospital Seattle, Washington, United States, 98105 Collapse << |
| NCT00551122 | Brain and Central Nervous Syst... more >>em Tumors Extragonadal Germ Cell Tumor Ovarian Cancer Testicular Germ Cell Tumor Collapse << | Phase 1 Phase 2 | Unknown | - | United Kingdom ... more >> Queen Elizabeth Hospital at University Hospital of Birmingham NHS Trust Recruiting Birmingham, England, United Kingdom, B15 2TH Contact: Michael H. Cullen, MD 0121-627-2444 Southampton General Hospital Recruiting Southampton, England, United Kingdom, SO16 6YD Contact: G. Mead, MD 44-23-8079-8639 Royal Marsden - Surrey Recruiting Sutton, England, United Kingdom, SM2 5PT Contact: Robert A. Huddart, MD 44-20-8661-3457 robert.huddart@icr.ac.uk Collapse << |
| NCT01074437 | - | - | Terminated(Insufficient enroll... more >>ment) Collapse << | - | - |
| NCT01248962 | Ovarian Cancer ... more >> Fallopian Tube Cancer Peritoneal Cancer Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT03301454 | Esophageal Cancer, Squamous Ce... more >>ll Collapse << | Phase 2 | Recruiting | February 2022 | France ... more >> Centre Hospitalier Universitaire Not yet recruiting Amiens, France, 80080 Contact: Vincent HAUTEFEUILLE, MD +33322088849 heutefeuille.vincent@chu-amiens.fr Principal Investigator: Vincent HAUTEFEUILLE, MD Centre Paul Papin Not yet recruiting Angers, France, 49055 Contact: Olivier CAPITAIN, MD +33240679900 olivier.capitain@ico.unicancer.fr Principal Investigator: Olivier CAPITAIN, MD Sub-Investigator: Véronique GUERIN-MEYER, MD Sub-Investigator: Julie VANBOCKSTAEL, MD Centre François Baclesse Not yet recruiting Caen, France, 14076 Contact: Marie-Pierre GALAIS, MD +33231455016 mp.galais@baclesse.fr Principal Investigator: Marie-Pierre GALAIS, MD Sub-Investigator: Françoise POLYCARPE-OSAER, MD Sub-Investigator: Aurélie PARZY, MD Sub-Investigator: Stéphane CORBINAIS, MD Centre Oscar Lambret Recruiting Lille, France, 59020 Contact: Farid EL HAJBI, MD +33320295266 f-elhajbi@o-lambret.fr Sub-Investigator: Frederik LAESTADIUS, MD Sub-Investigator: Diane PANNIER, MD Sub-Investigator: Natacha STERN, MD Sub-Investigator: Nicolas PENEL, PhD Sub-Investigator: Thomas RYCKEWAERT, MD Centre René Gauducheau Not yet recruiting Nantes, France, 44805 Contact: Olivier CAPITAIN, MD +33240679900 olivier.capitain@ico.unicancer.fr Sub-Investigator: Hélène SENELLART, MD Sub-Investigator: Sandrine HIRET, MD Principal Investigator: Olivier CAPITAIN, MD Sub-Investigator: Judith RAIMBOURG, MD Centre Armoricain de Radiothérapie, Imagerie médicale et Oncologie Not yet recruiting Plérin, France, 22190 Contact: Pierre-Luc ETIENNE, MD +33296752216 pl.etienne@cario-sante.fr Principal Investigator: Pierre-Luc ETIENNE, MD Sub-Investigator: Jérôme MARTIN-BABAU, MD Sub-Investigator: Dominique BESSON, MD Sub-Investigator: Anne-Claire HARDY-BESSARD, MD Centre Eugène Marquis Not yet recruiting Rennes, France, 35042 Contact: Julien EDELINE, Pr +33299253000 j.edeline@rennes.unicancer.fr Principal Investigator: Samuel LE SOURD, MD Sub-Investigator: Anais BODERE, MD Sub-Investigator: Julien EDELINE, PhD Sub-Investigator: Claire LARIBLE, MD Sub-Investigator: Fanny LE DU, MD Sub-Investigator: Astrid LIEVRE, MD Sub-Investigator: Marc PRACHT, MD Collapse << |
| NCT00855660 | Pharmacology, Clinical | Phase 1 | Completed | - | Germany ... more >> Köln, Nordrhein-Westfalen, Germany, 51147 Collapse << |
| NCT03710304 | Cesarean Section Complications | Not Applicable | Recruiting | June 1, 2020 | Egypt ... more >> Aswan University Recruiting Aswan, Egypt, 81528 Contact: hany f sallam, md 01092440504 ext 002 nahla.elsayed@aswu.ed.eg Collapse << |
| NCT00001910 | - | - | Completed | - | United States, Maryland ... more >> National Institute of Allergy and Infectious Diseases (NIAID) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01734759 | Healthy | Phase 1 | Completed | - | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00885417 | H. Pylori Infection | Phase 4 | Completed | - | Taiwan ... more >> National Taiwan University Hospital Taipei, Taiwan, 10002 Collapse << |
| NCT01032655 | Helicobacter Pylori Infection | Phase 4 | Completed | - | Taiwan ... more >> National Taiwan University Hospital Taipei, Taiwan, 10002 Collapse << |
| NCT01048229 | Early-stage Parkinson's Diseas... more >>e Collapse << | Phase 4 | Terminated(recruitment) | - | - |
| NCT00443963 | Dyspepsia | Phase 4 | Terminated(The Principal Inves... more >>tigator is no longer affiliated with the Study Site.) Collapse << | - | United States, District of Col... more >>umbia Washington Hospital Center 110 Irving St. NW, Washington, District of Columbia, United States, 20010 Collapse << |
| NCT03241602 | Decrease Post Laproscopy Shoul... more >>der and Abdominal Pain Collapse << | Phase 4 | Completed | - | Egypt ... more >> Faculty of medicine CAIRO UNIVERISTY Cairo, Egypt Collapse << |
| NCT01866449 | Glioblastoma Multiforme (GBM) ... more >>WHO Grade IV Collapse << | Phase 2 | Completed | - | Germany ... more >> Hämatologisch onkologische Praxis Augsburg, Germany, 86150 Stiftungsklinikum Mittelrhein GmbH Koblenz, Germany, 56068 Lars Bullinger, MD Ulm, Germany, 89081 Collapse << |
| NCT00985608 | Helicobacter Pylori Infection | Phase 2 | Unknown | September 2009 | Italy ... more >> Institute of Internal Medicine - Catholic University Recruiting Rome, Italy, 00168 Contact: Giovanni Cammarota, MD 0039-06-30155948 gcammarota@rm.unicatt.it Principal Investigator: Giovanni Cammarota, MD Collapse << |
| NCT03644550 | Mesothelioma | Phase 2 | Recruiting | February 28, 2023 | United States, Maryland ... more >> National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT03715881 | Non-Arteritic Anterior Ischemi... more >>c Optic Neuropathy (NAION) Collapse << | Phase 2 | Recruiting | March 1, 2019 | Iran, Islamic Republic of ... more >> Ophthalmic Research Center Recruiting Tehran, Iran, Islamic Republic of Contact: Homayon Nikkhah, MD 009822591616 labbafi@hotmail.com Collapse << |
| NCT00037570 | Peptic Ulcer Hemorrhage | Phase 2 | Completed | - | United States, California ... more >> Los Angeles, California, United States, 90073 San Diego, California, United States, 62103-8401 United States, Ohio Cincinnati, Ohio, United States, 45267-0595 United States, Pennsylvania Philadelphia, Pennsylvania, United States, 19140 United States, Texas Houston, Texas, United States, 77030-4211 United States, Virginia Norfolk, Virginia, United States, 23507 Collapse << |
| NCT02570529 | Pancreatic Ductal Adenocarcino... more >>ma Collapse << | Phase 4 | Unknown | February 2018 | - |
| NCT00285688 | - | - | Unknown | December 2008 | Thailand ... more >> Rajavithi Hospital Recruiting Ratchathewi, Bangkok, Thailand, 10400 Contact: Piyathida Harnsomboonrana, M.D. 6618423854 pboontee@yahoo.com Collapse << |
| NCT00199342 | Stage IV Melanoma | Phase 1 Phase 2 | Terminated(Occurrence of 2 dos... more >>e-limiting toxicities at the initial dose level (60 mg/m2) in Part I.) Collapse << | - | United States, Alabama ... more >> Comprehensive Cancer Center- University of Alabama at Birmingham Birmingham, Alabama, United States, 35294 United States, Florida H. Lee Moffitt Cancer Center Tampa, Florida, United States, 22612 Collapse << |
| NCT01028807 | Early Feeding ... more >> Bowel Anastomosis Collapse << | Not Applicable | Completed | - | Mexico ... more >> Hospital Infantil de Mexico Mexico Df, DF, Mexico, 06720 Collapse << |
| NCT01083537 | Ovarian Cancer ... more >> Peritoneal Cancer Fallopian Tube Cancer Bowel Obstruction Collapse << | Phase 1 Phase 2 | Terminated(Slow accrual) | - | Canada, Ontario ... more >> Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT02169856 | Postoperative Nausea and Vomit... more >>ing Collapse << | Not Applicable | Completed | - | Pakistan ... more >> Benazir Bhutto Hospital Rawalpindi. Rawalpindi, Punjab, Pakistan, 46000 Collapse << |
| NCT02441894 | Prostate Cancer | Phase 4 | Completed | - | Japan ... more >> Investigational Site Number 392004 Chuo-ku, Chiba, Japan Investigational Site Number 392008 Kita-gun, Japan Investigational Site Number 392007 Kobe-shi, Hyogo, Japan Investigational Site Number 392005 Nagakute-shi, Aichi, Japan Investigational Site Number 392006 Osaka Sayama-shi, Osaka, Japan Investigational Site Number 392001 Shinjuku-ku, Tokyo, Japan Investigational Site Number 392002 Yokohama-shi, Kanagawa, Japan Investigational Site Number 392009 Yokohama-shi, Japan Collapse << |
| NCT02441673 | Post Anaesthetic Shivering | Phase 2 | Not yet recruiting | June 2019 | - |
| NCT02999633 | T-cell Type Acute Leukemia-Pre... more >>cursor T-lymphoblastic Lymphoma/Leukaemia Collapse << | Phase 2 | Terminated(Due to an unsatisfa... more >>ctory benefit/risk ratio, as specified in & 14.8.1 of the protocol, Sanofi decided to stop enrollment and terminate ACT14596 prematurely) Collapse << | - | United States, Georgia ... more >> Investigational Site Number 8400002 Atlanta, Georgia, United States, 30342 United States, New Jersey Investigational Site Number 8400003 Hackensack, New Jersey, United States, 07601 United States, Texas Investigational Site Number 8400001 Houston, Texas, United States, 77030 Finland Investigational Site Number 2460001 Helsinki, Finland, 00029 France Investigational Site Number 2500005 Nantes Cedex 01, France, 44093 Investigational Site Number 2500001 Paris Cedex 10, France, 75475 Investigational Site Number 2500004 Pessac, France, 33600 Investigational Site Number 2500002 Pierre Benite, France, 69310 Hungary Investigational Site Number 3480001 Budapest, Hungary, 1083 Investigational Site Number 3480003 Budapest, Hungary, 1097 Investigational Site Number 3480002 Debrecen, Hungary, 4032 Italy Investigational Site Number 3800001 Bergamo, Italy, 24127 Investigational Site Number 3800004 Brescia, Italy, 25123 Lithuania Investigational Site Number 4400001 Vilnius, Lithuania, 08661 Russian Federation Investigational Site Number 6430003 Moscow, Russian Federation, 117198 Investigational Site Number 6430004 Moscow, Russian Federation, 125167 Investigational Site Number 6430001 Moscow, Russian Federation, 129301 Collapse << |
| NCT00930670 | Coronary Artery Disease | Phase 4 | Completed | - | Canada, Quebec ... more >> Institut Universitaire de Cardiologie et de Pneumologie de Quebec Quebec City, Quebec, Canada, G1V4G5 Collapse << |
| NCT00247715 | Dyspepsia Gas... more >>trointestinal Diseases Collapse << | Not Applicable | Completed | - | Netherlands ... more >> Radboud University Nijmegen Medical Center Nijmegen, Gelderland, Netherlands, 6500 HB Maastricht University Maastricht, Limburg, Netherlands, 6200 MB UMC Utrecht Utrecht, Netherlands Collapse << |
| NCT02004197 | Helicobacter Pylori [H. Pylori... more >>] as the Cause of Diseases Collapse << | Phase 2 Phase 3 | Completed | - | Pakistan ... more >> clinical trial was conducted in high risk areas of Pakistan such as Karachi, Bahawalpur, Islamabad and Rawalpindi at Shifa-Ul-Mulk Memorial Hospital, Hamdrad University Karachi, Bahawalpur Victoria Hospital and Nawaz Salik Hospital respectively Karachi, Pakistan Shifa Ul MUlk Memorial Hospital Karachi, Pakistan Collapse << |
| NCT01209455 | Poisoning | Not Applicable | Unknown | September 2012 | United Kingdom ... more >> Clinical Research Facility, Royal Infirmary of Edinburgh Recruiting Edinburgh, Midlothian, United Kingdom, EH16 4SA Principal Investigator: Euan A Sandilands, MRCP BSc Collapse << |
| NCT03619408 | - | - | Not yet recruiting | July 2020 | - |
| NCT02966665 | Chronic Obstructive Pulmonary ... more >>Disease Pulmonary Artery Hypertension Heart Failure Hypertension Collapse << | Phase 1 | Recruiting | August 2020 | United States, Utah ... more >> George E Wahlen VA Medical Center Recruiting Salt Lake City, Utah, United States, 84132 Contact: Russell Richardson, Ph.D. 801-582-1565 r.richardson@hsc.utah.edu Contact: Ashley Nelson, MD 801-582-1565 ext 4127 ash.nelson@hsc.utah.edu Collapse << |
| NCT01274130 | Healthy Adult | Not Applicable | Completed | - | Korea, Republic of ... more >> Severance Hospital Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT02555852 | - | - | Completed | - | Canada, Quebec ... more >> Lady Davis Institute for Medical Research, Jewish General Hospital Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT02123498 | Eustachian Tube Dysfunction ... more >> Laryngopharyngeal Reflux Collapse << | Phase 4 | Withdrawn(IRB not approved) | - | United States, California ... more >> 11234 Anderson Street Loma Linda, California, United States, 92354 1895 Orange Tree Lane, Suite 102 Redlands, California, United States, 92374 Collapse << |
| NCT00131248 | Gastroesophageal Reflux | Phase 3 | Completed | - | United States, Texas ... more >> Memorial Hermann Children's Hospital Houston, Texas, United States, 77030 Collapse << |
| NCT00223691 | Autonomic Failure ... more >> Orthostatic Hypotension Collapse << | Phase 1 | Completed | - | United States, Tennessee ... more >> Vanderbilt University Nashville, Tennessee, United States, 37232 Collapse << |
| NCT00131248 | - | - | Completed | - | - |
| NCT02258815 | Neuroblastoma Recurrent | Phase 2 | Unknown | December 2015 | Austria ... more >> University Hospital Graz Recruiting Graz, Austria, 8036 Contact: Wolfgang Schwinger, MD, PhD +43 31638512605 wolfgang.schwinger@medunigraz.at Principal Investigator: Wolfgang Schwinger, MD, PhD St. Anna Childrens Hospital Recruiting Vienna, Austria, 1090 Contact: Ruth Ladenstein, MD, PhD 43 1 40470 3250 ruth.ladenstein@ccri.at Principal Investigator: Ruth Ladenstein, MD, PhD Germany University Hospital Greifswald Recruiting Greifswald, Germany, 17475 Contact: Holger Lode, MD, PhD +49 3834866301 lode@uni-greifswald.de Principal Investigator: Holger Lode, MD, PhD University Hospital Tuebingen Recruiting Tuebingen, Germany, 72076 Contact: Peter Lang, MD, PhD +4970712983781 peter.lang@med.uni-tuebingen.de Principal Investigator: Peter Lang, MD, PhD Collapse << |
| NCT00161200 | Esophageal Metaplasia | Phase 3 | Terminated(slow accrual) | - | United States, New Jersey ... more >> Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08901 Collapse << |
| NCT01261546 | Parapneumonic Pleural Effusion... more >> Empyema Collapse << | Phase 2 | Completed | - | Spain ... more >> Hospital Carlos Haya Malaga, Andalucia, Spain Complejo Hospitalario Toledo Toledo, Castilla La Mancha, Spain Hospital Principe de Asturias Alcalá de Henares, Madrid, Spain Hospital Universitario de Getafe Getafe, Madrid, Spain Hospital Infanta Sofia San Sebastián de los Reyes, Madrid, Spain, 28014 Hospital Infantil La Paz Madrid, Spain, 28037 Hospital Ramón y Cajal Madrid, Spain Hospital Universitario Gregorio Marañón Madrid, Spain Collapse << |
| NCT00220909 | Patients Undergoing Elective C... more >>oronary Artery Bypass Graft Collapse << | Phase 4 | Terminated(Recruitment slow) | - | United States, Pennsylvania ... more >> Temple University School of Medicine Philadelphia, Pennsylvania, United States, 19140 Collapse << |
| NCT01332630 | - | - | Completed | - | - |
| NCT02703896 | Pulmonary Aspiration of Gastri... more >>c Contents Collapse << | Phase 4 | Completed | - | - |
| NCT00030992 | - | - | Completed | - | - |
| NCT01332630 | Breast Cancer | Phase 2 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00000964 | HIV Infections | Phase 1 | Completed | - | United States, Maryland ... more >> Johns Hopkins Adult AIDS CRS Baltimore, Maryland, United States, 21205 Collapse << |
| NCT00590928 | Critically Ill Patients ... more >> Indication for Stress Ulcer Prophylaxis Collapse << | Phase 4 | Completed | - | Austria ... more >> Medical University of Vienna, Department of Medicine III, ICU Vienna, Austria, 1090 Collapse << |
| NCT03667703 | Congenital Heart Disease ... more >> Upper Gastrointestinal Bleeding Stress Ulcer Infection Collapse << | Not Applicable | Not yet recruiting | June 30, 2021 | United States, Massachusetts ... more >> Boston Children's Hospital Not yet recruiting Boston, Massachusetts, United States, 02115 Contact: Kimberly I Mills, MD 617-355-7866 kimberly.mills@cardio.chboston.org Contact: Ben D Albert, MD 617-355-7866 ben.albert@childrens.harvard.edu Collapse << |
| NCT00030992 | Renal Cell Carcinoma | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00585351 | Stroke | Not Applicable | Completed | - | United States, Iowa ... more >> University of Iowa Iowa City, Iowa, United States, 52242 Collapse << |
| NCT03140033 | Hemorrhage Postpartum | Phase 2 | Recruiting | May 2017 | Egypt ... more >> Ain shams maternity hospital Recruiting Cairo, Egypt Contact: Mohamed Fathy, M.B.B.Ch 00201099984100 Mohamedfathy31085@gmail.com Contact: AMR Helmy, MD.MRCOG 00201227900014 am_helmy77@hotmail.com Collapse << |
| NCT00405119 | Gastroesophageal Reflux Diseas... more >>e Reflux, Gastroesophageal Collapse << | Phase 2 | Completed | - | Australia, New South Wales ... more >> GSK Investigational Site Randwick, New South Wales, Australia, 2031 Australia, Queensland GSK Investigational Site Herston, Queensland, Australia, 4006 Collapse << |
| NCT03583567 | Protamine Adverse Reaction | Phase 4 | Recruiting | December 1, 2020 | Thailand ... more >> Siriraj Hospital Recruiting Bangkok, Thailand, 10700 Contact: Sirilak Suksompoong, MD ssuksompong5@gmail.com Collapse << |
| NCT03468777 | Healthy | Phase 1 | Terminated(The study has been ... more >>terminated due to pending data analysis) Collapse << | - | Belgium ... more >> Clinical Pharmacology Unit Merksem, Belgium, 2170 Collapse << |
| NCT03011463 | Pharmacokinetics ... more >> Inhibition Enzyme Drug Interaction Potentiation Collapse << | Phase 1 | Completed | - | - |
| NCT00702871 | Ventilator Associated Pneumoni... more >>a Etiological Organisms Antimicrobial Drug Susceptibility Pattern Stress Ulcer Prophylaxis Collapse << | Phase 4 | Completed | - | India ... more >> Maulana Azad Medical College and Lok Nayak Hospital Delhi, India, 110002 Collapse << |
| NCT01976078 | - | - | Completed | - | United States, California ... more >> Children's Hospital Los Angeles Los Angeles, California, United States, 90027 Collapse << |
| NCT02700087 | Laryngomalacia ... more >> Acid Reflux Stridor Collapse << | Not Applicable | Withdrawn | February 2017 | United States, California ... more >> Lucile Packard Children's Hospital Stanford Palo Alto, California, United States, 94304 Collapse << |
| NCT02733640 | Antiplatelet Effect | Not Applicable | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seongnam, Gyeonggi-do, Korea, Republic of, 463-707 Collapse << |
| NCT00668317 | - | - | Terminated(Primary care physic... more >>ians began prescribing antacid therapy for chronic cough) Collapse << | - | - |
| NCT01682408 | Pharmacokinetics | Phase 1 | Completed | - | United States, Kansas ... more >> Overland Park, Kansas, United States Collapse << |
| NCT00585351 | - | - | Completed | - | - |
| NCT02172417 | Healthy | Phase 1 | Completed | - | - |
| NCT00633412 | NSAID Associated Gastric Ulcer... more >>s Collapse << | Phase 3 | Completed | - | - |
| NCT00839306 | Gastroesophageal Reflux Diseas... more >>e (GERD) Collapse << | Phase 3 | Completed | - | United States, Illinois ... more >> Associates, Ltd. Moline, Illinois, United States, 61265 Midwest Clinical Moline, Illinois, United States, 61265 Research Moline, Illinois, United States, 61265 Moline, Illinois, United States, 61265 Collapse << |
| NCT00839306 | - | - | Completed | - | - |
| NCT01392755 | Healthy Volunteer | Phase 1 | Completed | - | United States, Kansas ... more >> Lenexa, Kansas, United States, 66219 Collapse << |
| NCT01908257 | Healthy | Phase 1 | Completed | - | United States, California ... more >> Cypress, California, United States, 90630 Collapse << |
| NCT00668317 | Cough | Phase 3 | Terminated(Primary care physic... more >>ians began prescribing antacid therapy for chronic cough) Collapse << | - | United Kingdom ... more >> Clinical Trials Unit , Cardiovascular and respiratory studies, Castle Hill Hospital Cottingham, East Yorkshire, United Kingdom, HU16 5JQ Collapse << |
| NCT00401752 | Gastric Ulcer | Phase 3 | Completed | - | China, Fujian ... more >> Research Site Fuzhou, Fujian, China China, Guangdong Research Site Guangzhou, Guangdong, China China, Hubei Research Site Wuhan, Hubei, China China, Jiangsu Research Site Suzhou, Jiangsu, China China, Liaoning Research Site Shenyang, Liaoning, China China, Shanxi Research Site Xi'an, Shanxi, China China Research Site Beijing, China Research Site Shanghai, China Hong Kong Research Site Hong Kong, Hong Kong Collapse << |
| NCT00401752 | - | - | Completed | - | - |
| NCT00633672 | NSAID Associated Gastric Ulcer... more >>s Collapse << | Phase 3 | Completed | - | - |
| NCT00838526 | - | - | Completed | - | - |
| NCT00002106 | HIV Infections | Phase 2 | Completed | - | United States, California ... more >> Pacific Oaks Med Group Sherman Oaks, California, United States, 91403 United States, Florida Community Research Initiative Coral Gables, Florida, United States, 33146 United States, North Carolina Duke Univ Med Ctr Durham, North Carolina, United States, 27710 United States, Texas Houston Clinical Research Network Houston, Texas, United States, 77006 Collapse << |
| NCT03145012 | Cancer | Phase 4 | Not yet recruiting | April 30, 2019 | Canada, Nova Scotia ... more >> Nova Scotia Health Authority Not yet recruiting Halifax, Nova Scotia, Canada, B3H 1V7 Contact: Lisa Barrett, MD lisa.barrett@nshealth.ca Contact: Jill Moore jill.moore@nshealth.ca Collapse << |
| NCT01538797 | Reflux Oesophagitis | Phase 1 | Completed | - | United Kingdom ... more >> Hammersmith Medicines Research London, United Kingdom, NW10 7EW Collapse << |
| NCT02170792 | Healthy | Phase 1 | Completed | - | - |
| NCT00838526 | Gastroesophageal Reflux Diseas... more >>e (GERD) Collapse << | Phase 3 | Completed | - | United States, Illinois ... more >> Ltd. Moline, Illinois, United States, 61265 Midwest Clinical Moline, Illinois, United States, 61265 Research Associates Moline, Illinois, United States, 61265 Moline, Illinois, United States, 61265 Collapse << |
| NCT01737840 | - | - | Completed | - | - |
| NCT00247130 | Peptic Ulcers | Phase 4 | Withdrawn(Superiority of iv om... more >>eprazole to ranitidine has already been proven by others.) Collapse << | - | Japan ... more >> Department of Internal Medicine, Keio University School of Medicine Tokyo, Japan, 1608582 Collapse << |
| NCT01539655 | Medullary Thyroid Cancer | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT02197143 | Dyspepsia | Phase 4 | Completed | - | Turkey ... more >> Pamukkale Universty Denizli, Turkey, 20000 Collapse << |
| NCT00527878 | - | - | Terminated(Failure to enroll a... more >>dequate patient numbers due to small number of eligible patients) Collapse << | - | - |
| NCT01737840 | Dyspepsia | Phase 4 | Completed | - | Turkey ... more >> Akdeniz University Antalya, Turkey, 07050 Collapse << |
| NCT01896557 | - | - | Completed | - | - |
| NCT01131702 | Healthy | Phase 1 | Completed | - | Canada, Quebec ... more >> Anapharm Inc. Montreal, Quebec, Canada, G1V 2K8 Collapse << |
| NCT00527878 | JOB's Syndrome ... more >> Hyper-IgE Recurrent Infection Syndrome Immune Deficiency Collapse << | Phase 2 | Terminated(Failure to enroll a... more >>dequate patient numbers due to small number of eligible patients) Collapse << | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02195804 | Healthy | Phase 1 | Completed | - | - |
| NCT01896557 | Coronary Artery Disease ... more >> Drug Interaction Potentiation Collapse << | Phase 4 | Completed | - | - |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
