CAS No.: 443913-73-3
凡德他尼 Catalog No. CSN12186
Synonyms: ZD6474;CH 331;Zactima
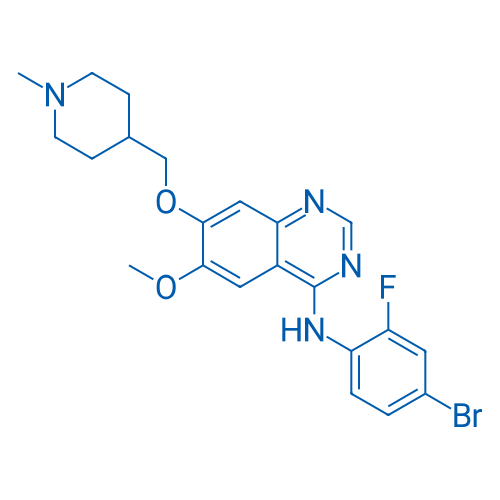
Vandetanib is a potent inhibitor of VEGFR2 with IC50 of 40 nM.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 VEGFR2
IC50:40nM- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
动物研究 剂量 Rat: 30.8 mg/kg[5] (p.o.) Mice: 25 mg/kg[6] (o.g., i.p.), 12.5 mg/kg- 50 mg/kg[4] (p.o.)
给药途径 p.o., o.g., i.p.
动物药代数据 Animal Rats[7] Dose 5 mg/kg (male) Administration i.v. or p.o. F 91.8% (p.o.) T1/2 15.6 h (i.v.)
19.6 h (p.o.)AUC0→36h 2920 ng·h/ml (i.v.)
2680 ng·h/ml (p.o.)Tmax 3.0 h (p.o.) Cmax 142 ng/ml (p.o.) AUC 3560 ng·h/ml (i.v.)
3530 ng·h/ml (p.o.)- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02142036 Metastatic Cancer Phase 2 Active, not recruiting January 2022 Norway ... more >> Akershus University Hospital Lillestrøm, Norway, 1478 The Norwegian Radium Hospital Oslo, Norway, 0379 Collapse << NCT03413176 - - Completed - France ... more >> AP-HP, Pitié-Salpêtrière Hospital, Department of Pharmacology, CIC-1421, Pharmacovigilance Unit, INSERM. Paris, France, 75013 Collapse << NCT02788201 Urothelial Carcinoma ... more >> Bladder Cancer Urinary Bladder Neoplasms Collapse << Phase 2 Recruiting July 1, 2020 United States, Maryland ... more >> National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << - 更多
- 参考文献
- [1] Vitagliano D, De Falco V, et al. The tyrosine kinase inhibitor ZD6474 blocks proliferation of RET mutant medullary thyroid carcinoma cells. Endocr Relat Cancer. 2010 Nov 30;18(1):1-11.
- [2] Wedge SR, Ogilvie DJ, et al. ZD6474 inhibits vascular endothelial growth factor signaling, angiogenesis, and tumor growth following oral administration. Cancer Res. 2002 Aug 15;62(16):4645-55.
- [3] Inoue K, Torimura T, et al. Vandetanib, an inhibitor of VEGF receptor-2 and EGF receptor, suppresses tumor development and improves prognosis of liver cancer in mice. Clin Cancer Res. 2012 Jul 15;18(14):3924-33.
- [4] Yoshikawa D, Ojima H, et al. Vandetanib (ZD6474), an inhibitor of VEGFR and EGFR signalling, as a novel molecular-targeted therapy against cholangiocarcinoma. Br J Cancer. 2009;100(8):1257-66.
- [5] Attwa MW, Kadi AA, et al. Identification and characterization of in vivo, in vitro and reactive metabolites of vandetanib using LC-ESI-MS/MS. Chem Cent J. 2018 Sep 24;12(1):99.
- [6] Pozo K, Zahler S, et al. Preclinical characterization of tyrosine kinase inhibitor-based targeted therapies for neuroendocrine thyroid cancer. Oncotarget. 2018 Dec 28;9(102):37662-37675.
- [7] ZD6474
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 443913-73-3 | 储存条件 |
|
|||||||||||||
| 分子式 | C22H24BrFN4O2 | 运输 | 蓝冰 | |||||||||||||
| 分子量 | 475.35 | 别名 | ZD6474;CH 331;Zactima;AZD-6474;CHEBI:38942;Zactim;Caprelsa;凡德他尼 | |||||||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| 201T | 2.5 μM | Function Assay | 48 h | inhibits phospho-MAPK following EGF | 22258476 |
| 201T | 1/5/10 μM | Function Assay | 48 h | blocks the phosphorylation of Akt induced by VEGFC | 22258476 |
| 211H | - | Growth Inhibition Assay | 72 h | IC50=2.2 μM | 18364248 |
| 273T | 2.5 μM | Function Assay | 48 h | inhibits phospho-MAPK following EGF | 22258476 |
| 8505C | - | Growth Inhibition Assay | 72 h | IC50=7.56 ± 1.13 μM | 21220477 |
| A-431 | - | Growth Inhibition Assay | 72 h | GI50=2.4 ± 0.3 μM | 24681205 |
| A549 | 2.5 μM | Function Assay | 48 h | inhibits phospho-MAPK following EGF | 22258476 |
| ACC2 | 0-10 μM | Growth Inhibition Assay | 72 h | inhibits cell growth in a dose dependent manner | 18698025 |
| ACC2 | 0-10 μM | Apoptosisi Assay | 72 h | induces apoptosis dose dependently | 18698025 |
| ACC3 | 0-10 μM | Growth Inhibition Assay | 72 h | inhibits cell growth in a dose dependent manner | 18698025 |
| ACC3 | 0-10 μM | Apoptosisi Assay | 72 h | induces apoptosis dose dependently | 18698025 |
| ACCM | 0-10 μM | Growth Inhibition Assay | 72 h | inhibits cell growth in a dose dependent manner | 18698025 |
| ACCM | 0-10 μM | Apoptosisi Assay | 72 h | induces apoptosis dose dependently | 18698025 |
| C643 | - | Growth Inhibition Assay | 72 h | IC50=3.65 ± 1.22 μM | 21220477 |
| C666-1 | 0.1-25.6 μM | Growth Inhibition Assay | 48 h | IC50=23.4 μM | 17631646 |
| C666-1 | 0.1-25.6 μM | Growth Inhibition Assay | 72 h | IC50=4.86 μM | 17631646 |
| C666-1 | 6 μM | Function Assay | 24 h | delays G0/G1 cell cycle progression | 17631646 |
| CNE-1 | 0.1-25.6 μM | Growth Inhibition Assay | 48 h | IC50=3.6 μM | 17631646 |
| CNE-1 | 0.1-25.6 μM | Growth Inhibition Assay | 72 h | IC50=2.3 μM | 17631646 |
| CNE-1 | 6 μM | Function Assay | 24 h | delays G0/G1 cell cycle progression | 17631646 |
| CNE-2 | 0.1-25.6 μM | Growth Inhibition Assay | 48 h | IC50=6.2 μM | 17631646 |
| CNE-2 | 0.1-25.6 μM | Growth Inhibition Assay | 72 h | IC50=3.6 μM | 17631646 |
| CNE-2 | 6 μM | Function Assay | 24 h | delays G0/G1 cell cycle progression | 17631646 |
| CRL1749 | 0-20 μM | Growth Inhibition Assay | 24 h | inhibits cell growth in a dose dependent manner | 19220256 |
| EHMES-1 | - | Growth Inhibition Assay | 72 h | IC50=10.6 μM | 18364248 |
| EHMES-10 | - | Growth Inhibition Assay | 72 h | IC50=0.3 μM | 18364248 |
| H1650 | - | Growth Inhibition Assay | - | IC50=3.5±1.2 μM | 23274758 |
| H2052 | - | Growth Inhibition Assay | - | IC50=1.07±0.04 μM | 21970874 |
| H2052 | - | Growth Inhibition Assay | 72 h | IC50=8.0 μM | 18364248 |
| H2452 | - | Growth Inhibition Assay | - | IC50=3.52±1.13 μM | 21970874 |
| H2452 | - | Growth Inhibition Assay | 72 h | IC50=5.5 μM | 18364248 |
| H28 | - | Growth Inhibition Assay | - | IC50=0.32±0.07 μM | 21970874 |
| H28 | - | Growth Inhibition Assay | 72 h | IC50=1.8 μM | 18364248 |
| HAK1-B | 1/5/10 μM | Function Assay | 1 h | suppresses EGFR phosphorylation | 22611027 |
| HMEpC | 1 nM-100 μM | Growth Inhibition Assay | 48 h | inhibits cell growth in a dose dependent manner | 24138843 |
| HT1376 | 0-20 μM | Growth Inhibition Assay | 24 h | inhibits cell growth in a dose dependent manner | 19220256 |
| HTB3 | 0-20 μM | Growth Inhibition Assay | 24 h | inhibits cell growth in a dose dependent manner | 19220256 |
| HTB9 | 0-20 μM | Growth Inhibition Assay | 24 h | inhibits cell growth in a dose dependent manner | 19220256 |
| Hth104 | - | Growth Inhibition Assay | 72 h | IC50=±16.98 ± NA μM | 21220477 |
| Hth7 | - | Growth Inhibition Assay | 72 h | IC50=9.66 ± 0.38 μM | 21220477 |
| Hth74 | - | Growth Inhibition Assay | 72 h | IC50=8.56 ± 1.01 μM | 21220477 |
| Hth83 | - | Growth Inhibition Assay | 72 h | IC50=3.30 ± 0.66 μM | 21220477 |
| HuH-7 | - | Growth Inhibition Assay | 72 h | IC50 = 9.4 μmol/L | 22611027 |
| HUVECs | - | Growth Inhibition Assay | 72 h | IC50 = 7.1 μmol/L | 22611027 |
| HUVECs | 1/5/10 μM | Function Assay | 1 h | significantly inhibits VEGFR-2 phosphorylation | 22611027 |
| J82 | 0-20 μM | Growth Inhibition Assay | 24 h | inhibits cell growth in a dose dependent manner | 19220256 |
| Jurkat | - | Growth Inhibition Assay | 72 h | GI50=1.5 ± 0.2 μM | 24681205 |
| K-562 | - | Growth Inhibition Assay | 72 h | GI50=1.8 ± 0.1 μM | 24681205 |
| KYN-2 | - | Growth Inhibition Assay | 72 h | IC50 = 8.1 μmol/L | 22611027 |
| MCF-7 | 1 nM-100 μM | Growth Inhibition Assay | 48 h | inhibits cell growth in a dose dependent manner | 24138843 |
| MDA-MB-231 | 1 nM-100 μM | Growth Inhibition Assay | 48 h | inhibits cell growth in a dose dependent manner | 24138843 |
| MDA-MB-468 | 1 nM-100 μM | Growth Inhibition Assay | 48 h | inhibits cell growth in a dose dependent manner | 24138843 |
| MSTO-211H | - | Growth Inhibition Assay | - | IC50=1.42±0.03 μM | 21970874 |
| NCTC-2544 | - | Growth Inhibition Assay | 72 h | GI50=4.6 ± 0.3 μM | 24681205 |
| PCI-15B | 0-6 μM | Growth Inhibition Assay | 72 h | inhibits cell growth in a dose dependent manner | 22307735 |
| PCI-15B | 0-10 μM | Function Assay | 24 h | inhibits the activation of the EGFR tyrosine kinase and also decreases the expression of phosphorylated forms of the downstream signaling elements, STAT3 and MAPK | 22307735 |
| PCI-15B | 1 μM | Function Assay | 24 h | downregulates VEGF production | 22307735 |
| PCI-15B | - | Invasion Assay | 24 h | EC50=558 nM | 22307735 |
| PCI-37A | 0-6 μM | Growth Inhibition Assay | 72 h | inhibits cell growth in a dose dependent manner | 22307735 |
| PCI-37A | 1 μM | Function Assay | 24 h | downregulates VEGF production | 22307735 |
| PCI-37A | - | Invasion Assay | 24 h | EC50=1695 nM | 22307735 |
| PCI-37B | 0-6 μM | Growth Inhibition Assay | 72 h | inhibits cell growth in a dose dependent manner | 22307735 |
| PCI-37B | - | Invasion Assay | 24 h | EC50=1726 nM | 22307735 |
| RT4 | 0-20 μM | Growth Inhibition Assay | 24 h | inhibits cell growth in a dose dependent manner | 19220256 |
| SCC-25 | 0-6 μM | Growth Inhibition Assay | 72 h | inhibits cell growth in a dose dependent manner | 22307735 |
| SCC-25 | - | Invasion Assay | 24 h | EC50=10 nM | 22307735 |
| SH-SY5Y | 0.625-20 μM | Growth Inhibition Assay | 48 h | inhibits cell growth in a dose dependent manner | 24399074 |
| SH-SY5Y | 5/10/20 μM | Apoptosisi Assay | 48 h | induces apoptosis dose dependently | 24399074 |
| SH-SY5Y | 5/10/20 μM | Function Assay | 48 h | induces G1 phase cell cycle arrest | 24399074 |
| SH-SY5Y | 1/5/10 μM | Function Assay | 48 h | inhibits RET phosphorylation | 24399074 |
| SH-SY5Y | 5/10 μM | Function Assay | 48 h | inhibits human NB cell migration | 24399074 |
| SH-SY5Y | 5/10 μM | Function Assay | 48 h | inhibits human NB cell invasion | 24399074 |
| SH-SY5Y | 5 μM | Function Assay | 24/48/72 h | suppresses the expression of CXCR4 and MMP14 mRNA | 24399074 |
| SH-SY5Y | 5 μM | Function Assay | 48/72 h | suppresses expression of the CXCR4 and MMP14 protein | 24399074 |
| SK-N-SH | 0.625-20 μM | Growth Inhibition Assay | 48 h | inhibits cell growth in a dose dependent manner | 24399074 |
| SK-N-SH | 5/10/20 μM | Apoptosisi Assay | 48 h | induces apoptosis dose dependently | 24399074 |
| SK-N-SH | 5/10/20 μM | Function Assay | 48 h | induces G1 phase cell cycle arrest | 24399074 |
| SK-N-SH | 1/5/10 μM | Function Assay | 48 h | inhibits RET phosphorylation | 24399074 |
| SK-N-SH | 5/10 μM | Function Assay | 48 h | inhibits human NB cell migration | 24399074 |
| SK-N-SH | 5/10 μM | Function Assay | 48 h | inhibits human NB cell invasion | 24399074 |
| SK-N-SH | 5 μM | Function Assay | 24/48/72 h | suppresses the expression of CXCR4 and MMP14 mRNA | 24399074 |
| SK-N-SH | 5 μM | Function Assay | 48/72 h | suppresses expression of the CXCR4 and MMP14 protein | 24399074 |
| SN179 | 500 nM | Function Assay | 16 h | increases CXCR4 expression significantly | 25676691 |
| SN179 | 500 nM | Function Assay | 16 h | enhances the CXCL12 directed migration | 25676691 |
| SN179 | 500 nM | Function Assay | 16 h | increases basal migration | 25676691 |
| SN186 | 500 nM | Function Assay | 16 h | increases CXCR4 expression significantly | 25676691 |
| SUP | 0-20 μM | Growth Inhibition Assay | 24 h | inhibits cell growth in a dose dependent manner | 19220256 |
| SW1736 | - | Growth Inhibition Assay | 72 h | IC50=9.05 ± 0.55 μM | 21220477 |
| T24 | 0-20 μM | Growth Inhibition Assay | 24 h | inhibits cell growth in a dose dependent manner | 19220256 |
| T-47-D | 1 nM-100 μM | Growth Inhibition Assay | 48 h | inhibits cell growth in a dose dependent manner | 24138843 |
| U251 | 2/4/8 μℳ | Function Assay | 6/12/24 h | increases the LC3-II level in a time-dependent and dose-dependent manner | 23799852 |
| U251 | 4 μℳ | Function Assay | 2/6/12 h | suppresses basal levels of phosphorylation of S6 (S235/236), 4E-BP1 (T37/46), and Akt (S473) in a time-dependent manner | 23799852 |
| U87MG | 2/4/8 μℳ | Function Assay | 6/12/24 h | increases the LC3-II level in a time-dependent and dose-dependent manner | 23799852 |
| U87MG | 4 μℳ | Function Assay | 2/6/12 h | suppresses basal levels of phosphorylation of S6 (S235/236), 4E-BP1 (T37/46), and Akt (S473) in a time-dependent manner | 23799852 |
| UM-22A | 0-6 μM | Growth Inhibition Assay | 72 h | inhibits cell growth in a dose dependent manner | 22307735 |
| UM-22A | 0-10 μM | Function Assay | 24 h | inhibits the activation of the EGFR tyrosine kinase and also decreases the expression of phosphorylated forms of the downstream signaling elements, STAT3 and MAPK | 22307735 |
| UM-22A | 1 μM | Function Assay | 24 h | downregulates VEGF production | 22307735 |
| UM-22A | - | Invasion Assay | 24 h | EC50=0.3 nM | 22307735 |
| UM-22B | 0-6 μM | Growth Inhibition Assay | 72 h | inhibits cell growth in a dose dependent manner | 22307735 |
| UM-22B | 0-10 μM | Function Assay | 24 h | inhibits the activation of the EGFR tyrosine kinase and also decreases the expression of phosphorylated forms of the downstream signaling elements, STAT3 and MAPK | 22307735 |
| UM-22B | - | Invasion Assay | 24 h | EC50=2424 nM | 22307735 |
| ZR-75-1 | 1 nM-100 μM | Growth Inhibition Assay | 48 h | inhibits cell growth in a dose dependent manner | 24138843 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02142036 | Metastatic Cancer | Phase 2 | Active, not recruiting | January 2022 | Norway ... more >> Akershus University Hospital Lillestrøm, Norway, 1478 The Norwegian Radium Hospital Oslo, Norway, 0379 Collapse << |
| NCT03413176 | - | - | Completed | - | France ... more >> AP-HP, Pitié-Salpêtrière Hospital, Department of Pharmacology, CIC-1421, Pharmacovigilance Unit, INSERM. Paris, France, 75013 Collapse << |
| NCT02788201 | Urothelial Carcinoma ... more >> Bladder Cancer Urinary Bladder Neoplasms Collapse << | Phase 2 | Recruiting | July 1, 2020 | United States, Maryland ... more >> National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT00499850 | Advanced Colorectal Carcinoma | Phase 1 | Completed | - | Australia ... more >> Research Site East Melbourne, Australia Research Site Footscray, Australia Research Site Heidelberg, Australia Research Site Parkville, Australia Collapse << |
| NCT02117167 | Non-small Cell Lung Cancer Met... more >>astatic Collapse << | Phase 2 | Recruiting | February 2022 | France ... more >> Centre Hospitalier Henri Duffau Recruiting Avignon, France Contact: Nicolas CLOAREC, MD nicolas.cloarec@ch-avignon.fr Principal Investigator: Nicolas CLOAREC, MD Centre Hospitalier Universitaire de Besancon - Hopital Jean Minjoz Recruiting Besancon, France Contact: Virginie WESTEEL, MD virginie.westeel@univ.fcomte.fr Principal Investigator: Virginie WESTEEL, MD Hôpital Avicenne Recruiting Bobigny, France Contact: Boris DUCHEMANN, MD boris.duchemann@aphp.fr Principal Investigator: Boris DUCHEMANN, MD Institut Bergonié Recruiting Bordeaux, France Contact: François Chomy, MD f.chomy@bordeaux.unicancer.fr Principal Investigator: François Chomy, MD Hôpital Ambroise Paré Recruiting Boulogne Billancourt, France Contact: Etienne GIROUX LEPRIEUR, MD etienne.giroux-leprieur@aphp.fr Principal Investigator: Etienne GIROUX LEPRIEUR, MD Hospices Civils de Lyon- Hôpital Louis Pradel Recruiting Bron, France Contact: Nicolas Girard, MD nicolas.girard@chu-lyon.fr Principal Investigator: Nicolas Girard, MD Centre François Baclesse Recruiting Caen, France Contact: Radj Gervais, MD r.gervais@baclesse.unicancer.fr Principal Investigator: Radj Gervais, MD CHU Caen Recruiting Caen, France Contact: Gérard Zalcman, MD zalcman-g@chu-caen.fr Principal Investigator: Gérard Zalcman, MD Chu de Caen - Hopital Cote de Nacre Recruiting Caen, France Contact: Jeannick MADELAINE, MD madelaine-j@chu-caen.fr Principal Investigator: Jeannick MADELAINE, MD Hôpital Louis Pasteur Recruiting Chartres, France Contact: Claire Lethrosne, MD clethrosne@ch-chartres.fr Principal Investigator: Claire Lethrosne, MD centre Jean Perrin Recruiting Clermont-Ferrand, France Contact: Xavier Durando, MD xavier.durando@cjp.fr Principal Investigator: Xavier Durando, MD CHU Clermont Ferrand - Hôpital Gabriel Montpied Recruiting Clermont-Ferrand, France Contact: Henri Janicot, MD hjanicot@chu-clermontferrand.fr Principal Investigator: Henri Janicot, MD Hopitaux Civils de Colmar Recruiting Colmar, France Contact: Lionel MOREAU, MD lionel.moreau@ch-colmar.fr Principal Investigator: Lionel MOREAU, MD Centre Hopsitalier Intercommunal de Créteil Recruiting Créteil, France Contact: Isabelle Monnet, MD isabelle.monnet@chicreteil.fr Principal Investigator: Isabelle Monnet, MD Centre Georges François Leclerc Recruiting Dijon, France Contact: Bruno Coudert, MD bcoudert@cgfl.fr Principal Investigator: Bruno Coudert, MD CHU Grenoble Recruiting Grenoble, France Contact: Denis Moro-Sibilot, MD Dmoro-Sibilot@chu-grenoble.fr Principal Investigator: Denis Moro-Sibilot, MD Chd Vendee Recruiting La Roche Sur Yon, France Contact: Tifenn L'HARIDON, MD Principal Investigator: Tifenn L'HARIDON, MD CH du Mans Recruiting Le Mans, France Contact: Olivier Molinier, MD omolinier@ch-lemans.fr Principal Investigator: Olivier Molinier, MD Centre Oscar Lambret Recruiting Lille, France Contact: Eric Dansin, MD e-dansin@o-lambret.fr Principal Investigator: Eric Dansin, MD CHRU de Lille Recruiting Lille, France Contact: Alexis Cortot, MD Alexis.CORTOT@CHRU-LILLE.FR Principal Investigator: Alexis Cortot, MD Centre Léon Bérard Recruiting Lyon, France Contact: Maurice Pérol, MD maurice.perol@lyon.unicancer.fr Principal Investigator: Maurice Pérol, MD Hôpital Nord Recruiting Marseille, France Contact: Fabrice Barlesi, MD fabrice.barlesi@ap-hm.fr Principal Investigator: Fabrice Barlési, MD Institut Paoli Calmettes Recruiting Marseille, France Contact: Anne MADROSZYK, MD madroszyka@ipc.unicancer.fr Principal Investigator: Anne MADROSZYK, MD Institut de cancérologie de l'Ouest Recruiting Nantes, France Contact: Jaafar Bennouna, MD jaafar.bennouna@ico.unicancer.fr Principal Investigator: Jaafar Bennouna, MD Centre Antoine Lacassagne Recruiting Nice, France Contact: Josiane OTTO, MD josiane.otto@nice.unicancer.fr Principal Investigator: Josiane OTTO, MD Chr Orleans Recruiting Orleans, France Contact: Hugues MOREL, MD hugues.morel@chr-orleans.fr Contact: , MD Principal Investigator: Hugues MOREL AH-HP Hôpital Saint Louis Recruiting Paris, France Contact: Damien Pouessel, MD damien.pouessel@sls.aphp.fr Principal Investigator: Damien Pouessel, MD AP-HP Hôpital Cochin Recruiting Paris, France Contact: Jeanne Chapron, MD jeanne.chapron@cch.aphp.fr Principal Investigator: Jeanne Chapron, MD AP-HP Hôpital Européen Georges Pompidou Withdrawn Paris, France AP-HP Hôpital Tenon Recruiting Paris, France Contact: Marie Wislez, MD marie.wislez@tnn.aphp.fr Principal Investigator: Marie Wislez, MD Institut Curie Recruiting Paris, France Contact: Catherine Daniel, MD catherine.daniel@curie.fr Principal Investigator: Catherine Daniel, MD Centre Hospitalier de Pau Recruiting PAU, France Contact: Aldo RENAULT, MD aldo.renault@ch-pau.fr Principal Investigator: Aldo RENAULT, MD Centre Hospitalier Lyon Sud Recruiting Pierre Bénite, France Contact: Jean-Pierre Souquet, MD pierre-jean.souquet@chu-lyon.fr Principal Investigator: Jean-Pierre Souquet, MD CHR Pontchailloux Withdrawn Rennes, France Chru Strasbourg - Nouvel Hopital Civil Recruiting Strasbourg, France Contact: Philippe BARTHELEMY, MD Principal Investigator: Philippe BARTHELEMY, MD CHI de Toulon - Hôpital Sainte-Musse Recruiting Toulon, France Contact: Xavier TCHIKNAVORIAN, MD Principal Investigator: Xavier TCHIKNAVORIAN, MD CHU Toulouse -Hôpital Larrey Recruiting Toulouse, France Contact: Julien Mazières, MD mazieres.j@chu-toulouse.fr Principal Investigator: Julien Mazières, MD Hôpital Bretonneau Recruiting Tours, France Contact: Eric Pichon, MD e.pichon@chu-tours.fr Principal Investigator: Eric Pichon, MD Gustave Roussy Recruiting Villejuif, France Contact: Benjamin Besse, MD Benjamin.BESSE@gustaveroussy.fr Principal Investigator: Benjamin Besse, MD Sub-Investigator: Jean-Charles Soria, MD Collapse << |
| NCT02299999 | Metastatic Breast Cancer | Phase 2 | Recruiting | December 2022 | France ... more >> Institut de Cancérologie de l'Ouest/Paul Papin Recruiting Angers, France Contact: Mario CAMPONE, MD mario.campone@ico.unicancer.fr Principal Investigator: Mario CAMPONE, MD Institut Sainte-Catherine Recruiting Avignon, France Contact: Alice Mege, MD a.mege@isc84.org Principal Investigator: Alice Mege, MD Polyclinique Bordeaux Nord Aquitaine Recruiting Bordeaux, France, 33077 Contact: Nadine DOHOLLOU, MD n.dohollou@bordeauxnord.com Contact: Nadine DOHOLLOU, MD Institut Bergonié Recruiting Bordeaux, France Contact: Hervé Bonnefoi, MD h.bonnefoi@bordeaux.unicancer.fr Principal Investigator: Hervé Bonnefoi, MD Centre François Baclesse Recruiting Caen, France Contact: Christelle Lévy, MD c.levy@baclesse.unicancer.fr Principal Investigator: Christelle Lévy, MD Centre Jean Perrin Recruiting Clermont-Ferrand, France Contact: Marie-Ange Mouret Reynier, MD marie-ange.mouret-reynier@cjp.fr Principal Investigator: Marie-Ange Mouret Reynier, MD Ch Alpes Leman Not yet recruiting Contamine Sur Arve, France, 74130 Contact: Carol ALLIOT, MD calliot@ch-alpes-leman.fr Contact: Carol ALLIOT, MD Centre Georges François Leclerc Recruiting Dijon, France, 21079 Contact: Nicolas Isambert, MD nisambert@cgfl.fr Principal Investigator: Nicolas Isambert, MD Chd Vendee Recruiting La Roche-sur-Yon, France, 85925 Contact: Tifenn L'Haridon, MD tifenn.lharidon@chd-vendee.fr Contact: Tifenn L'Haridon, MD Centre Oscar Lambret Recruiting Lille, France Contact: Nuria KOTECKI, MD Principal Investigator: Nuria KOTECKI, MD Chu Dupuytren Not yet recruiting Limoges, France, 87000 Contact: Laurence VENAT-BOUVET, MD laurence.venat-bouvet@chu-limoges.fr Contact: Laurence VENAT-BOUVET, MD Hopital Privé Jean Mermoz Not yet recruiting Lyon, France, 69008 Contact: Olfa DERBEL, MD o.derbelmermoz@gmail.com Contact: Olfa DERBEL, MD Centre Hospitalier Lyon Sud Recruiting Lyon, France Contact: Benoit You, MD Principal Investigator: Benoit You, MD Centre Léon Bérard Recruiting Lyon, France Contact: Thomas Bachelot, MD thomas.bachelot@lyon.unicancer.fr Principal Investigator: Thomas Bachelot, MD Institut Paoli Calmettes Recruiting Marseille, France Contact: Anthony Gonçalves, MD goncalvesa@ipc.unicancer.fr Principal Investigator: Anthony Gonçalves, MD Institut Régional du Cancer Montpellier Val d'Aurelle Recruiting Montpellier, France Contact: William Jacot, MD william.jacot@icm.unicancer.fr Principal Investigator: William Jacot, MD Centre Alexis Vautrin Recruiting Nancy, France Contact: Elisabeth Luporsi, MD e.luporsi@nancy.unicancer.fr Principal Investigator: Elisabeth Luporsi, MD Institut de Cancérologie de l'Ouest/ René Gauducheau Recruiting Nantes, France Contact: Mario Campone, MD mario.campone@ico.unicancer.fr Principal Investigator: Mario Campone, MD Centre Antoine Lacassagne Recruiting Nice, France Contact: Jean-Marc Ferrero, MD jean-marc.ferrero@nice.unicancer.fr Principal Investigator: Jean-Marc Ferrero, MD Institut Curie Recruiting Paris, France Contact: Marie-Paule Sablin, MD mariepaule.sablin@curie.fr Principal Investigator: Marie-Paule Sablin, MD Centre Eugène Marquis Recruiting Rennes, France Contact: Claudia Lefeuvre-Plesse, MD c.lefeuvre@rennes.unicancer.fr Principal Investigator: Claudia Lefeuvre-Plesse, MD Centre Henri Becquerel Recruiting Rouen, France Contact: Jean-Christophe Théry, MD Principal Investigator: Jean-Christophe Théry, MD Institut Curie Recruiting Saint-Cloud, France Contact: Florence COUSSY, MD florence.coussy@curie.fr Principal Investigator: Florence COUSSY, MD Hopitaux Universitaire de Strasbourg - Hopital Civil Recruiting Strasbourg, France Contact: Philippe BARTHELEMY, MD philippe.barthelemy@chru-strasbourg.fr Principal Investigator: Philippe BARTHELEMY, MD Hopitaux Du Leman Recruiting Thonon-les-Bains, France, 74200 Contact: Francesco DEL PIANO, MD f-delpiano@ch-hopitauxduleman.fr Contact: Francesco DEL PIANO, MD Institut Claudius Regaud Recruiting Toulouse, France Contact: Florence Dalenc, MD dalenc.florence@iuct-oncopole.fr Principal Investigator: Florence Dalenc, MD Gustave Roussy Recruiting Villejuif, France Contact: Monica Arnedos, MD Monica.ARNEDOS@gustaveroussy.fr Principal Investigator: Monica Arnedos, MD Sub-Investigator: Fabrice André, MD Collapse << |
| NCT00496509 | Colorectal Cancer | Phase 1 | Completed | - | Germany ... more >> Research Site Freiberg, Germany Collapse << |
| NCT00290537 | - | - | Terminated(Accrual was subopti... more >>mal and increasing the number of patients was not feasible.) Collapse << | - | - |
| NCT01838642 | Thyroid Neoplasms | Phase 2 | Terminated(Recruiting halted p... more >>rematurely and will not resume. New study to open soon.) Collapse << | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01838642 | - | - | Terminated(Recruiting halted p... more >>rematurely and will not resume. New study to open soon.) Collapse << | - | - |
| NCT02638428 | Relapsed Pediatric Solid Tumor... more >> Refractory Pediatric Solid Tumor Relapsed Pediatric AML Refractory Pediatric AML Collapse << | Phase 2 | Recruiting | December 2023 | Korea, Republic of ... more >> Samsung Medical Center Recruiting Seoul, Korea, Republic of Contact: Ki Woong Sung 82-2-3410-3529 kwsped@skku.edu Principal Investigator: Ki Woong Sung Collapse << |
| NCT00290537 | Lung Cancer | Phase 2 | Terminated(Accrual was subopti... more >>mal and increasing the number of patients was not feasible.) Collapse << | - | United States, Texas ... more >> University of Texas M.D.Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00533169 | Neuroblastoma | Phase 1 | Terminated(Lack of enrollment.... more >>) Collapse << | - | United States, Texas ... more >> U.T.M.D. Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00252746 | Non Small Cell Lung Carcinoma | Phase 2 | Completed | - | Japan ... more >> Research Site Matsuyama, Ehime, Japan Research Site Isehara, Kanagawa, Japan Research Site Osakasayama, Osaka, Japan Research Site Toyonaka, Osaka, Japan Research Site Sunto-gun, Shizuoka, Japan Research Site Okayama, Japan Research Site Tokyo, Japan Collapse << |
| NCT02013089 | Gastrointestinal Cancers | Not Applicable | Unknown | December 2017 | China, Guangdong ... more >> Gastrointestinal Hospital, Sun Yat-sen University Recruiting Guangzhou, Guangdong, China, 510655 Contact: Yanghong Deng, PhD 008613925106525 13925106525@163.com Principal Investigator: Yanhong Deng, PhD Collapse << |
| NCT00066313 | Lung Cancer | Phase 2 | Completed | - | Canada, Alberta ... more >> Tom Baker Cancer Centre - Calgary Calgary, Alberta, Canada, T2N 4N2 Cross Cancer Institute at University of Alberta Edmonton, Alberta, Canada, T6G 1Z2 Canada, British Columbia British Columbia Cancer Agency - Centre for the Southern Interior Kelowna, British Columbia, Canada, V1Y 5L3 Fraser/Valley Cancer Centre at British Columbia Cancer Agency Surrey, British Columbia, Canada, V3V 1Z2 British Columbia Cancer Agency - Vancouver Cancer Centre Vancouver, British Columbia, Canada, V5Z 4E6 Canada, New Brunswick Moncton Hospital Moncton, New Brunswick, Canada, E1C 6ZB Saint John Regional Hospital Saint John, New Brunswick, Canada, E2L 4L2 Canada, Ontario Margaret and Charles Juravinski Cancer Centre Hamilton, Ontario, Canada, L8V 5C2 Cancer Centre of Southeastern Ontario at Kingston General Hospital Kingston, Ontario, Canada, K7L 5P9 Ottawa Hospital Regional Cancer Centre - General Campus Ottawa, Ontario, Canada, K1H 8L6 St. Catharines General Hospital at Niagara Health System St. Catharines, Ontario, Canada, L2R 5K3 Northwestern Ontario Regional Cancer Care at Thunder Bay Regional Health Sciences Centre Thunder Bay, Ontario, Canada, P7B 6V4 Toronto East General Hospital Toronto, Ontario, Canada, M4C 3E7 Toronto Sunnybrook Regional Cancer Centre at Sunnybrook and Women's College Health Sciences Centre Toronto, Ontario, Canada, M4N 3M5 Mount Sinai Hospital - Toronto Toronto, Ontario, Canada, M5G 1X5 Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Windsor Regional Cancer Centre at Windsor Regional Hospital Windsor, Ontario, Canada, N8W 2X3 Canada, Quebec Hopital Notre- Dame du CHUM Montreal, Quebec, Canada, H2L 4M1 McGill Cancer Centre at McGill University Montreal, Quebec, Canada, H2W 1S6 L'Hopital Laval Ste-Foy, Quebec, Canada, G1V 4G5 Canada, Saskatchewan Saskatoon Cancer Centre at the University of Saskatchewan Saskatoon, Saskatchewan, Canada, S7N 4H4 Collapse << |
| NCT00410189 | Lung Cancer | Phase 2 | Completed | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00745732 | Non-Small Cell Lung Cancer | Phase 1 | Terminated(Sponsor withdrew su... more >>pport.) Collapse << | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00659438 | Prostate Cancer | Phase 2 | Completed | - | France ... more >> Research Site Bordeaux Cedex, France Research Site Creteil, France Research Site Paris, France Research Site Reims Cedex, France Research Site Villejuif, France Collapse << |
| NCT02239952 | Cancer High-g... more >>rade Glioma Collapse << | Not Applicable | Recruiting | January 2019 | Netherlands ... more >> VU University Medical Center Recruiting Amsterdam, Noord-Holland, Netherlands, 1081 HV Contact: H.M.W Verheul, MD, PhD +31 (0)20 4444321 h.verheul@vumc.nl Collapse << |
| NCT00613054 | Glioblastoma ... more >>Gliosarcoma Collapse << | Phase 1 | Completed | - | United States, North Carolina ... more >> Duke University Health System Durham, North Carolina, United States, 27710 Collapse << |
| NCT00358956 | Thyroid Cancer | Phase 2 | Completed | - | United States, Arkansas ... more >> Research Site Little Rock, Arkansas, United States United States, Massachusetts Research Site Boston, Massachusetts, United States Australia Research Site St Leonards, Australia Canada, Quebec Research Site Sherbrooke, Quebec, Canada Italy Research Site Pisa, Italy Netherlands Research Site Utrecht, Netherlands Romania Research Site Bucharest, Romania Spain Research Site Madrid, Spain Switzerland Research Site Basel, Switzerland Collapse << |
| NCT00358956 | - | - | Completed | - | - |
| NCT00496665 | Metastatic Breast Cancer | Phase 1 | Completed | - | United States, Massachusetts ... more >> Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Massachusetts General Hospital Boston, Massachusetts, United States, 02215 Collapse << |
| NCT00410189 | - | - | Completed | - | - |
| NCT00404924 | Non-Small-Cell Lung Carcinoma | Phase 3 | Completed | - | - |
| NCT00659438 | - | - | Completed | - | - |
| NCT00811369 | Metastatic Breast Cancer | Phase 2 | Completed | - | Canada, Alberta ... more >> Cross Cancer Institute Edmonton, Alberta, Canada, T6G 1Z2 Canada, British Columbia British Columbia Cancer Agency - Vancouver Centre Vancouver, British Columbia, Canada, V5Z 4E6 Canada, Nova Scotia QE II Health Sciences Centre Halifax, Nova Scotia, Canada, B3H 1V7 Canada, Ontario Juravinski Cancer Centre Hamilton, Ontario, Canada, L8V 5C2 Grand River Regional Cancer Centre Kitchener, Ontario, Canada, N2G 1G3 RSM Durham Regional Cancer Centre Oshawa, Ontario, Canada, L1G 2B9 Ottawa Hospital Cancer Centre Ottawa, Ontario, Canada, K1H 8L6 Regional Cancer Program of the Hôpital régional de Sudbury Regional Hospital Sudbury, Ontario, Canada, P3E 5J1 Odette Cancer Centre - Sunnybrook Health Sciences Centre Toronto, Ontario, Canada, M4N 3M5 St. Michael's Hospital Toronto, Ontario, Canada, M5B 1W8 Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Canada, Quebec Centre Hospitalier De L'Universite De Montreal - Hotel Dieu Montreal, Quebec, Canada, H2W 1T7 Canada, Saskatchewan Saskatoon Cancer Centre Saskatoon, Saskatchewan, Canada, S7N 4H4 Collapse << |
| NCT00454116 | Colorectal Cancer | Phase 2 | Completed | - | United States, Michigan ... more >> Research Site Ann Arbor, Michigan, United States United States, New York Research Site New York, New York, United States United States, Tennessee Research Site Nashville, Tennessee, United States United States, Utah Research Site Salt Lake City, Utah, United States Argentina Research Site Buenos Aires, Argentina Research Site Ramos Mejia, Argentina Research Site Rosario, Argentina Research Site Santa Fe, Argentina Research Site Vicente Lopez, Argentina Korea, Republic of Research Site Seoul, Korea, Republic of Norway Research Site Bergen, Norway Research Site Oslo, Norway Research Site Stavanger, Norway Spain Research Site A Coruna, Spain Research Site Barcelona, Spain Research Site Jaen, Spain Research Site Lerida, Spain Research Site Lleida, Spain United Kingdom Research site Belfast, Northern Ireland, United Kingdom Research Site Aberdeen, United Kingdom Research Site Leicester, United Kingdom Research Site Manchester, United Kingdom Collapse << |
| NCT00454116 | - | - | Completed | - | - |
| NCT00364351 | Non Small Cell Lung Cancer | Phase 3 | Completed | - | - |
| NCT00498797 | Prostate Cancer ... more >> Metastatic Hormone Refractory Collapse << | Phase 2 | Completed | - | Brazil ... more >> Research site Rio de Janeiro, Brazil Research Site Sao Paulo, Brazil Germany Research Site Hamburg, Germany Research Site Hannover, Germany Research Site Kassel, Germany Research Site Tubingen, Germany Hungary Research Site Budapest, Hungary South Africa Research Site Bloemfontein, South Africa Research Site Cape Town, South Africa Sweden Research Site Umea, Sweden Research Site Uppsala, Sweden Collapse << |
| NCT03630120 | Thyroid Cancer ... more >> Thyroid Cancer, Medullary Differentiated Thyroid Cancer Papillary Thyroid Cancer Follicular Thyroid Cancer Poorly Differentiated Thyroid Gland Carcinoma Collapse << | Phase 2 | Recruiting | December 2023 | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Recruiting Tampa, Florida, United States, 33612 Collapse << |
| NCT00364351 | - | - | Completed | - | - |
| NCT00494481 | - | - | Completed | - | - |
| NCT00404924 | - | - | Completed | - | - |
| NCT00494481 | Advanced Breast Cancer | Phase 2 | Completed | - | Hungary ... more >> Research Site Budapest, Hungary Research Site Pécs, Hungary South Africa Research SIte Bloemfontein, South Africa Research Site Cape Town, South Africa Research Site Observatory, South Africa Spain Research Site Baracaldo, Spain Research SIte Lérida, Spain Research Site Zaragoza, Spain Sweden Research Site Umeå, Sweden Research Site Uppsala, Sweden Research Site Västerås, Sweden Taiwan Research Site Taipei, Taiwan Collapse << |
| NCT00410761 | Thyroid Cancer | Phase 3 | Active, not recruiting | December 31, 2019 | - |
| NCT00410761 | - | - | Active, not recruiting | - | - |
| NCT00450138 | Head and Neck Cancer | Phase 1 | Completed | - | United States, Colorado ... more >> Research Site Denver, Colorado, United States United States, Illinois Research Site Chicago, Illinois, United States United States, Texas Research Site Dallas, Texas, United States Research Site Houston, Texas, United States Collapse << |
| NCT00681798 | Pancreatic Cancer | Phase 1 | Completed | - | Switzerland ... more >> Research Site St Gallen, SG, Switzerland Research Site Bellinzona, Switzerland Collapse << |
| NCT00507091 | Metastatic Co... more >>lorectal Adenocarcinoma Collapse << | Phase 1 | Completed | - | Belgium ... more >> Research Site Gent, Belgium United Kingdom Research Site Belfast, United Kingdom Research Site Manchester, United Kingdom Collapse << |
| NCT00312377 | Non-small Cell Lung Cancer ... more >> Lung Cancer Collapse << | Phase 3 | Completed | - | - |
| NCT00503711 | Advanced Solid, Malignant Tumo... more >>rs Collapse << | Phase 1 | Completed | - | China ... more >> Research Site Guangzhou, China Collapse << |
| NCT00459121 | Lung Cancer | Phase 2 | Terminated | - | United States, Michigan ... more >> Barbara Ann Karmanos Cancer Institute Detroit, Michigan, United States, 48201-1379 Collapse << |
| NCT00312377 | - | - | Completed | - | - |
| NCT00418886 | Non Small Cell Lung Cancer ... more >> Lung Cancer Collapse << | Phase 3 | Active, not recruiting | December 2019 | - |
| NCT00601614 | Unspecified Adult Solid Tumor,... more >> Protocol Specific Collapse << | Phase 1 | Withdrawn(no enrollment) | - | - |
| NCT00418886 | - | - | Active, not recruiting | - | - |
| NCT00498797 | - | - | Completed | - | - |
| NCT00686036 | Prostate Cancer | Phase 2 | Terminated(Slow recruitment) | - | Canada, Alberta ... more >> Research Site Calgary, Alberta, Canada Research Site Edmonton, Alberta, Canada Canada, British Columbia Research Site Victoria, British Columbia, Canada Canada, Ontario Research Site Hamilton, Ontario, Canada Research Site Kingston, Ontario, Canada Research Site London, Ontario, Canada Research Site Toronto, Ontario, Canada Canada, Quebec Research Site Greenfield Park, Quebec, Canada Research Site Montreal, Quebec, Canada Research Site Sherbrooke, Quebec, Canada Canada Research Site Granby, Canada Collapse << |
| NCT00459121 | - | - | Terminated | - | - |
| NCT00686036 | - | - | Terminated(Slow recruitment) | - | - |
| NCT00752986 | Breast Cancer | Phase 2 | Terminated | - | Italy ... more >> Research Site Avellino, Italy Research Site Benevento, Italy Research Site Genova, Italy Research Site Milano, Italy Research Site Monserrato, Italy Research Site Napoli, Italy Research Site Palermo, Italy Research Site Prato, Italy Research Site Roma, Italy Research Site Trento, Italy Research Site Varese, Italy Collapse << |
| NCT00506051 | Carcinoma Non... more >>-Small Cell Lung Lung Cancer Collapse << | Phase 1 | Completed | - | - |
| NCT00752986 | - | - | Terminated | - | - |
| NCT00481845 | Breast Cancer | Phase 2 | Terminated(Low accrual) | - | United States, California ... more >> Stanford University School of Medicine Stanford, California, United States, 94305 Collapse << |
| NCT00937417 | Breast Cancer ... more >> Head and Neck Cancer Lung Cancer Prostate Cancer Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 1 | Withdrawn(Withdrawn because SW... more >>OG no longer pursuing this study at this time) Collapse << | September 2009 | - |
| NCT00481845 | - | - | Terminated(Low accrual) | - | - |
| NCT00734890 | Lung Cancer L... more >>ymphoma Lymphoproliferative Disorder Small Intestine Cancer Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> Warren Grant Magnuson Clinical Center - NCI Clinical Trials Referral Office Bethesda, Maryland, United States, 20892-1182 Collapse << |
| NCT00098345 | Thyroid Cancer | Phase 2 | Completed | - | United States, California ... more >> Research Site San Francisco, California, United States United States, Connecticut Research Site New Haven, Connecticut, United States United States, New York Research Site New York, New York, United States United States, North Carolina Research Site Durham, North Carolina, United States United States, Texas Research Site Houston, Texas, United States France Research Site Villejuif Cedex, France Collapse << |
| NCT01414426 | Lip and Oral Cavity Squamous C... more >>ell Carcinoma Oral Cavity Verrucous Carcinoma Precancerous Condition Collapse << | Phase 2 | Active, not recruiting | March 2018 | United States, Illinois ... more >> University of Chicago Chicago, Illinois, United States, 60637 Collapse << |
| NCT00508001 | - | - | Completed | - | - |
| NCT00597116 | Mesothelioma | Phase 2 | Terminated(Recruitment stopped... more >> according to early stopping rule (by protocol)) Collapse << | - | Germany ... more >> Research Site Essen, Germany Research Site Halle-Dolau, Germany Research Site Hamburg, Germany Research Site Heidelberg, Germany Switzerland Research Site Chur, Switzerland Research Site Zurich, Switzerland Collapse << |
| NCT00732745 | Adenocarcinoma of the Gastroes... more >>ophageal Junction Esophageal Cancer Collapse << | Phase 1 | Terminated(Lost funding for Ph... more >>ase II portion of study) Collapse << | - | United States, Ohio ... more >> Case Medical Center, University Hospitals Seidman Cancer Center, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106-5065 Collapse << |
| NCT00597116 | - | - | Terminated(Recruitment stopped... more >> according to early stopping rule (by protocol)) Collapse << | - | - |
| NCT00508001 | Carcinoma, Hepatocellular | Phase 2 | Completed | - | Taiwan ... more >> Research Site Tainan, Taiwan Research Site Taipei, Taiwan Research Site Taoyuan, Taiwan Collapse << |
| NCT00753714 | - | - | Completed | - | - |
| NCT00441142 | Glioblastoma Multiforme ... more >> Gliosarcoma Collapse << | Phase 1 Phase 2 | Active, not recruiting | January 2018 | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Dana Farber / Brigham and Women's Cancer Center Boston, Massachusetts, United States, 02115 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 United States, Michigan Henry Ford Hospital Detroit, Michigan, United States, 48202 United States, New York Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 United States, Pennsylvania University of Pittsburgh Cancer Institute Pittsburgh, Pennsylvania, United States, 15232-1305 United States, Virginia University of Virginia Charlottesville, Virginia, United States, 22908-4324 Collapse << |
| NCT00753675 | Biliary Tract Cancer ... more >> Gallbladder Cancer Cancer Of The Extrahepatic Bile Duct Ampullary Carcinoma Collapse << | Phase 2 | Completed | - | Italy ... more >> Research Site Brescia, BS, Italy Research Site Firenze, FI, Italy Research Site Genova, GE, Italy Research Site Milano, Mi, Italy Research Site Palermo, PA, Italy Research Site Aviano, PN, Italy Research Site Parma, PR, Italy Research Site Reggio Emilia, RE, Italy Research Site Ancona, Italy Research Site Livorno, Italy Research Site Napoli, Italy Research Site Pisa, Italy Research Site Ravenna, Italy Research Site Rho, Italy Research Site Torino, Italy Collapse << |
| NCT00441142 | - | - | Active, not recruiting | - | - |
| NCT00807170 | Non-small Cell Lung Cancer (NS... more >>CLC) Collapse << | Phase 1 | Terminated(Very slow recruitme... more >>nt) Collapse << | - | Netherlands ... more >> Research site Amsterdam, Netherlands Research site Groningen, Netherlands Research site Maastricht, Netherlands Collapse << |
| NCT00777179 | NSCLC | Phase 2 | Completed | - | Korea, Republic of ... more >> Research Site CheongJu, Republic of Korea, Korea, Republic of Research Site Gyeonggi-do, Republic of Korea, Korea, Republic of Research Site Gyeongsangnam-Do, Republic of Korea, Korea, Republic of Research Site Incheon, Republic of Korea, Korea, Republic of Research Site Seoul, Republic of Korea, Korea, Republic of Collapse << |
| NCT00098345 | - | - | Completed | - | - |
| NCT00500292 | - | - | Completed | - | - |
| NCT00777179 | - | - | Completed | - | - |
| NCT01757470 | - | - | Completed | - | Canada, Quebec ... more >> Research Site Montreal, Quebec, Canada Collapse << |
| NCT02268734 | - | - | Unknown | December 2014 | Italy ... more >> Fondazione IRCCS Istituto Nazionale dei Tumori, Milano Recruiting Milano, Italy, 20133 Contact: Laura Locati, MD +39 022390 ext 2765 laura.locati@istitutotumori.mi.it Principal Investigator: Laura Locati, MD Collapse << |
| NCT00753675 | - | - | Completed | - | - |
| NCT00720083 | Head and Neck Cancer | Phase 2 | Terminated(Withdrawal of drug ... more >>supply.) Collapse << | - | - |
| NCT00720083 | - | - | Terminated(Withdrawal of drug ... more >>supply.) Collapse << | - | - |
| NCT00667147 | Solid Tumors | Phase 1 | Completed | - | United States, California ... more >> University of Southern California (U.S.C.)/ Norris Comprehensive Cancer Center Los Angeles, California, United States, 90033 Collapse << |
| NCT00500292 | Colorectal Ca... more >>ncer Collapse << | Phase 2 | Completed | - | France ... more >> Research Site Lille Cedex, France Research Site Toulouse Cedex 9, France Hungary Research Site Budapest, Hungary Research Site Debrecen, Hungary Research Site Szeged, Hungary Korea, Republic of Research Site Seoul, Korea, Republic of Slovakia Research Site Bratislava, Slovakia Research Site Poprad, Slovakia Research Site Trnava, Slovakia Research Site Zilina, Slovakia Spain Research Site Hospitalet deLlobregat, Spain Research Site Oviedo, Spain Research Site Santander, Spain Taiwan Research Site Taipei, Taiwan Research Site Tao-Yuan, Taiwan Collapse << |
| NCT00872989 | Fallopian Tube Cancer ... more >> Ovarian Cancer Primary Peritoneal Cavity Cancer Collapse << | Phase 2 | Completed | - | - |
| NCT00872989 | - | - | Completed | - | - |
| NCT00822887 | Malignant Gliomas | Phase 1 | Completed | - | United States, Colorado ... more >> University of Colorado Health Science Center Aurora, Colorado, United States, 80045 Collapse << |
| NCT00753714 | Non Small Cell Lung Cancer | Phase 2 | Completed | - | Italy ... more >> Research Site Meldola, (fc), Italy Research Site Avellino, AV, Italy Research Site Bari, BA, Italy Research Site Treviglio, BG, Italy Research Site Bologna, BO, Italy Research Site Genova, GE, Italy Research Site Taormina, ME, Italy Research Site Milano, MI, Italy Research Site Perugia, PG, Italy Research Site Ravenna, RA, Italy Research Site Trento, TN, Italy Research Site Orbassano, TO, Italy Research Site Udine, UD, Italy Research Site Padova, Italy Research Site Roma, Italy Collapse << |
| NCT00995007 | - | - | Completed | - | - |
| NCT00687297 | Lung Cancer N... more >>on Small Cell Lung Cancer Collapse << | Phase 2 | Completed | - | - |
| NCT00683787 | Gastric Cancer | Phase 2 | Terminated(low accrual) | - | United States, New York ... more >> Roswell Park Cancer Institute Buffalo, New York, United States, 14263-0001 Collapse << |
| NCT00687297 | - | - | Completed | - | - |
| NCT00995007 | Glioblastoma Multiforme ... more >> Gliosarcoma Anaplastic Astrocytoma Anaplastic Oligodendroglioma Anaplastic Mixed Oligoastrocytoma Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00880334 | Transitional Cell Carcinoma ... more >> Bladder Cancer Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00880334 | - | - | Completed | - | - |
| NCT00532909 | Anal, Colon, and Rectal Cancer... more >>s Colorectal Neoplasms Colon/Rectal Cancer Collapse << | Phase 1 | Completed | - | United States, California ... more >> Stanford University School of Medicine Stanford, California, United States, 94305 Collapse << |
| NCT01372813 | Advanced Clear Cell Renal Carc... more >>inoma Collapse << | Phase 2 | Terminated(Terminated for insu... more >>fficient accrual.) Collapse << | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00537095 | - | - | Active, not recruiting | - | - |
| NCT00757692 | Prostate Cancer | Phase 2 | Completed | - | Canada, British Columbia ... more >> BC Cancer Agency - Centre for Southern Interior Kelowna, British Columbia, Canada, V1Y 5L3 BC Cancer Agency - Vancouver Centre Vancouver, British Columbia, Canada, V5Z 4E6 Canada, Ontario Juravinski Cancer Centre Hamilton, Ontario, Canada, L8V 5C2 Sunnybrook Health Sciences Centre Toronto, Ontario, Canada, M4N 3M5 Collapse << |
| NCT00683787 | - | - | Terminated(low accrual) | - | - |
| NCT00862836 | Ovarian Cancer | Phase 1 Phase 2 | Terminated(Lack of efficacy) | - | Germany ... more >> Research Site Ulm, Baden-Württemberg, Germany Research Site Wiesbaden, Hessen, Germany Research Site Essen, Nordrhein-Westfalen, Germany Research Site Kiel, Schleswig-Holstein, Germany Research Site Berlin, Germany Collapse << |
| NCT00537095 | Thyroid Neoplasms | Phase 2 | Active, not recruiting | December 31, 2019 | Belgium ... more >> Research Site Brussels, Belgium Denmark Research Site Odense, Denmark France Research Site Angers Cedex 9, France Research Site Angers Cedex, France Research Site Bordeaux Cedex, France Research Site Caen Cedex 5, France Research Site Caen Cedex, France Research Site Lyon Cedex, France Research Site Lyon, France Research Site Marseille Cedex 9, France Research Site Marseille Cedex, France Research Site Paris Cedex 10, France Research Site Paris Cedex 13, France Research Site Paris, France Research Site Villejuif Cedex, France Research Site Villejuif, France Norway Research Site Oslo, Norway Spain Research Site L'Hospitalet de Llobregat, Spain Research Site Madrid, Spain Sweden Research Site Lund, Sweden Research Site Stockholm, Sweden Switzerland Research Site Bern, Switzerland Collapse << |
| NCT01945762 | - | - | Recruiting | September 2019 | Belgium ... more >> Research Site Active, not recruiting Brussels, Belgium France Research Site Active, not recruiting Bordeaux, France Research Site Terminated Lyon, France Research Site Recruiting Paris, France Research Site Recruiting Villejuif, France Germany Research Site Recruiting Augsburg, Germany Research Site Active, not recruiting Essen, Germany Research Site Active, not recruiting Heidelberg, Germany Research Site Active, not recruiting Munchen, Germany Italy Research Site Recruiting Catania, Italy Research Site Recruiting MIlano, Italy Research Site Active, not recruiting Milano, Italy Research Site Active, not recruiting Naples, Italy Research Site Recruiting Padova, Italy Research Site Recruiting Pisa, Italy Research Site Recruiting Siena, Italy Luxembourg Research Site Active, not recruiting Luxembourg, Luxembourg Netherlands Research Site Active, not recruiting Groningen, Netherlands Research Site Recruiting Leiden, Netherlands Spain Research Site Recruiting Barcelona, Spain Research Site Active, not recruiting Madrid, Spain Research Site Recruiting Madrid, Spain Research Site Active, not recruiting Oviedo, Spain Research Site Terminated Sevilla, Spain United Kingdom Research Site Terminated Glasgow, United Kingdom Research Site Recruiting London, United Kingdom Research Site Terminated London, United Kingdom Research Site Active, not recruiting Manchester, United Kingdom Research Site Active, not recruiting Sheffield, United Kingdom Research Site Recruiting Sutton, United Kingdom Collapse << |
| NCT01372813 | - | - | Terminated(Terminated for insu... more >>fficient accrual.) Collapse << | - | - |
| NCT00862836 | - | - | Terminated(Lack of efficacy) | - | - |
| NCT00445549 | - | - | Terminated(The study closed be... more >>cause of inadequate early activity.) Collapse << | - | - |
| NCT00445549 | Ovarian Neoplasms ... more >> Fallopian Tube Neoplasms Peritoneal Neoplasms Collapse << | Phase 2 | Terminated(The study closed be... more >>cause of inadequate early activity.) Collapse << | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01191892 | Bladder Cancer ... more >> Transitional Cell Cancer of the Renal Pelvis and Ureter Ureter Cancer Urethral Cancer Collapse << | Phase 2 | Unknown | December 2014 | United Kingdom ... more >> Beatson West of Scotland Cancer Centre Recruiting Glasgow, Scotland, United Kingdom, G12 0YN Contact: Contact Person 44-141-301-7095 Rob.Jones@ggc.scot.nhs.uk Wales Cancer Trials Unit Recruiting Cardiff, Wales, United Kingdom, CF11 9LJ Contact: Contact Person 44-29-2019-6800 Ayr Hospital Recruiting Ayr, United Kingdom, KA66DX Contact: Margaret McKernan 01563 825749 margaret.mckernan@aaaht.scot.nhs.uk Principal Investigator: Jawaher Ansari Royal Bournemouth General Hospital Recruiting Bournemouth, United Kingdom, BH7 7DW Contact: Susannah Brock susannah.brock@poole.nhs.uk Principal Investigator: Susannah Brock Queens Hospital Recruiting Burton upon Trent, United Kingdom, DE13 0RB Contact: Pugazhenthi Pattu 01332 340131 pugazhenthi.pattu@nhs.net Principal Investigator: Pugazhenthi Pattu Velindre Hospital Recruiting City and County of Cardiff, United Kingdom, CF142TL Contact: Dr Lester, Jason 44-2920 196159 jason.lester2@wales.nhs.uk Principal Investigator: Jason Lester Western General Hospital Recruiting Edinburgh, United Kingdom, EH4 2XU Contact: Duncan McLaren duncan.mclaren@luht.scot.nhs.uk Principal Investigator: Duncan McLaren Calderdale Royal Infirmary Recruiting Halifax, United Kingdom, HX30PW Contact: Lisa Gledhill 01484 342925 lisa.gledhill@cht.nhs.uk Principal Investigator: Ursula Hofmann Huddersfield Royal Infirmary Recruiting Huddersfield, United Kingdom, HD3 3EA Contact: Uschi Hofmann 0484 847299 uschi.hofmann@cht.nhs.uk Principal Investigator: Uschi Hofmann The Royal Lancaster Infirmary Recruiting Lancaster, United Kingdom, LA1 4RP Contact: Alison Birtle 01524 583219 alison.birtle@lhtr.nhs.uk Principal Investigator: Alison Birtle St. James's University Hospital Recruiting Leeds, United Kingdom, LS9 7TF Contact: Satinder Jagdev 0113 2067645 satinder.jagdev@leedsth.nhs.uk Principal Investigator: Satinder Jagdev The Royal Free Hospital Not yet recruiting London, United Kingdom, NW3 2QG Principal Investigator: Maria Vilarino-Varela St Marys Hospital Not yet recruiting London, United Kingdom, W21NY Contact: Gillian Hornzee Gillian.Hornzee@imperial.nhs.uk Principal Investigator: Simon Stewart Charing Cross Hospital Not yet recruiting London, United Kingdom, W68RF Contact: Simon Stewart simon.stewart@imperial.nhs.uk Principal Investigator: Simon Stewart Christie Hospital Recruiting Manchester, United Kingdom, M20 4BX Contact: Tony Eliott 01619187214 tony.eliott@christie.nhs.uk Principal Investigator: Tony Eliott Mount Vernon Hospital Recruiting Northwood Middlesex, United Kingdom, HA6 2RN Contact: Peter Hoskin 01923 844533 peter.hoskin@nhs.net Principal Investigator: Peter Hoskin Churchill Hospital Recruiting Oxford, United Kingdom, OX37LJ Contact: Georgina Rogers patpandrewprotheroe@oncology.ox.ac.uk Principal Investigator: Andrew Protheroe Weston Park Hospital Recruiting Sheffield, United Kingdom, S102SJ Contact: Linda Evans 0114 226500 linda.evans@sth.nhs.uk Principal Investigator: Linda Evans Southampton General Hospital Recruiting Southampton, United Kingdom, S016 6YD Contact: Simon Crabb s.j.crabb@southampton.ac.uk Principal Investigator: Simon Crabb Royal Surrey County Hospital Recruiting Surrey, United Kingdom, GU27XX Contact: Katie Wood 01483 571122 katiewood@nhs.net Principal Investigator: Katie Wood The Royal Marsden Hospital Recruiting Surrey, United Kingdom, KT2 7QB Contact: Robert Huddart 020 8661 3457 robert.huddart@icr.ac.uk Principal Investigator: Robert Huddart Collapse << |
| NCT02015065 | GIST | Phase 2 | Active, not recruiting | August 31, 2023 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01601808 | Pancreatic Cancer | Phase 2 | Unknown | October 2013 | United Kingdom ... more >> Belfast City Hospital Not yet recruiting Belfast, United Kingdom, BT9 7AB Principal Investigator: Dr Martin Eatock The Royal Bournemouth Hospital Recruiting Bournemouth, United Kingdom, BH7 7DW Principal Investigator: Dr Tamas Hickish Bristol Haematology and Oncology Centre Not yet recruiting Bristol, United Kingdom, BS2 8ED Principal Investigator: Dr Stephen Falk Royal Surrey County Hospital Recruiting Guildford, United Kingdom, GU2 7XX Principal Investigator: Dr Gary Middleton Clatterbridge Centre for Oncology Recruiting Liverpool, United Kingdom, CH63 4JY Principal Investigator: Professor Daniel Palmer Royal Liverpool University Hospital Recruiting Liverpool, United Kingdom, L69 3GA Principal Investigator: Professor Daniel Palmer St Bartholomew's Hospital Not yet recruiting London, United Kingdom, EC1A 7BE Principal Investigator: Dr David Propper Guys & St Thomas Hospital Recruiting London, United Kingdom, SE1 9RT Principal Investigator: Dr Paul Ross Royal Marsden Hospital Not yet recruiting London, United Kingdom, SW3 6JJ Principal Investigator: Professor David Cunningham The Christie Hospital Recruiting Manchester, United Kingdom, M20 4BX Principal Investigator: Dr Richard Hubner James Cook University Hospital Recruiting Middlesbrough, United Kingdom, TS4 3BW Principal Investigator: Dr Nick Wadd Freeman Hospital Not yet recruiting Newcastle, United Kingdom, NE7 7DN Principal Investigator: Dr Fareeda Coxon Nottingham City Hospital Recruiting Nottingham, United Kingdom, NG5 1PB Principal Investigator: Dr Srinivasan Madhusudan Weston Park Hospital Recruiting Sheffield, United Kingdom, S10 2SJ Principal Investigator: Dr Jonathan Wadsley Collapse << |
| NCT00996723 | Diffuse Intrinsic Pontine Glio... more >>ma Collapse << | Phase 1 | Completed | - | United States, Tennessee ... more >> St. Jude Children's Research Hospital Memphis, Tennessee, United States, 38105 Collapse << |
| NCT00660725 | Advanced Incurable Solid Malig... more >>nancy Collapse << | Phase 1 | Completed | - | United States, Pennsylvania ... more >> University of Pittsburgh Cancer Institute / Hillman Cancer Center Pittsburgh, Pennsylvania, United States, 15232 Collapse << |
| NCT00514046 | Medullary Thyroid Carcinoma ... more >> Multiple Endocrine Neoplasia Type 2A Multiple Endocrine Neoplasia Type 2B Collapse << | Phase 1 Phase 2 | Active, not recruiting | July 27, 2022 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01941849 | Phaeochromocytoma ... more >> Paraganglioma Collapse << | Phase 1 | Withdrawn(Poor patient accrual... more >>) Collapse << | - | United Kingdom ... more >> Guy's and St Thomas' NHS Foundation Trust London, United Kingdom The Christie NHS Foundation Trust London, United Kingdom University College London Hospitals NHS Foundation Trust London, United Kingdom Collapse << |
| NCT01496313 | Thyroid Cancer | Phase 4 | Active, not recruiting | December 2019 | United States, Texas ... more >> Research Site Houston, Texas, United States Czech Republic Research Site Olomouc, Czech Republic Research Site Praha 5, Czech Republic India Research Site Bangalore Karnataka, India Research Site Vellore, India Israel Research Site Beer Sheva, Israel Research Site Haifa, Israel Research Site Jerusalem, Israel Research Site Petach Tikva, Israel Italy Research Site Catania, Italy Research Site Milano, Italy Research Site Palermo, Italy Research Site Pisa, Italy Research Site Roma, Italy Research Site Siena, Italy Research Site Torino, Italy Netherlands Research Site Groningen, Netherlands Research Site Leiden, Netherlands Poland Research Site Gliwice, Poland Research Site Warszawa, Poland Research Site Zgierz, Poland Russian Federation Research Site Saint Petersburg, Russian Federation United Kingdom Research Site Cardiff, United Kingdom Research Site Greater London, United Kingdom Research Site London, United Kingdom Research Site Tyne & Wear, United Kingdom Collapse << |
| NCT01004419 | Carcinoma, Non Small Cell Lung | Phase 1 | Withdrawn(Support for investig... more >>ational products has been withdrawn.) Collapse << | May 2011 | - |
| NCT00472017 | Brain and Central Nervous Syst... more >>em Tumors Collapse << | Phase 1 | Completed | - | United States, Tennessee ... more >> St. Jude Children's Research Hospital Memphis, Tennessee, United States, 38105 Collapse << |
| NCT02495103 | Renal Cell Carcinoma ... more >> Hereditary Leiomyomatosis and Renal Cell Cancer Papillary Renal Cell Carcinoma, Sporadic Collapse << | Phase 1 Phase 2 | Recruiting | May 1, 2022 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT02530411 | Neoplasms | Phase 2 | Recruiting | December 2020 | United Kingdom ... more >> Royal Bournemouth Hospital Recruiting Bournemouth, Dorset, United Kingdom, BH7 7DW Contact: Tamas Hickish, MA MD FRCP 01202 704789 tamashickish@rbch.nhs.uk Royal Devon and Exeter Hospital Recruiting Exeter, England, United Kingdom, EX2 5DW Contact: Anne Hong, MB ChB MRCP 01392 402118 anne.hong@nhs.net Royal Cornwall Hospital Recruiting Truro, Cornwall, England, United Kingdom, TR1 3LJ Contact: Duncan Wheatley, MBBS, FRCR 01872 258304 duncan.wheatley@rcht.cornwall.nhs.uk Velindre Cancer Center, Velindre Hospital Recruiting Cardiff, Wales, United Kingdom, CF14 2TL Contact: Robert Jones, MD PhD MCRCP 029 2061 5888 robert.jones14@wales.nhs.uk Royal United Hospital Bath Recruiting Bath, United Kingdom, BA1 3NG Contact: Mark Beresford, MD MRCP FRCR 01225 824797 mark.beresford@nhs.net Collapse << |
| NCT01586624 | Cancer Non Sm... more >>all Cell Lung Cancer Collapse << | Phase 1 | Active, not recruiting | March 2019 | United Kingdom ... more >> Cambridge Cancer Trials Centre (S4), Box 279, Addenbrooke's Hospital Hills Road, Cambridge, United Kingdom, CB2 0QQ The Christie NHS Foundation Trust Withington, Manchester, United Kingdom, M20 4BX Churchill Hospital Headington, Oxford, United Kingdom, OX3 7LJ The Freeman Hospital Newcastle, United Kingdom, NE7 7DN Collapse << |
| NCT00923247 | - | - | Terminated(Terminated due to s... more >>low accrual,primary endpoint reached & investigator left NIH.) Collapse << | - | - |
| NCT00923247 | Medullary Thyroid Carcinoma | Phase 1 Phase 2 | Terminated(Terminated due to s... more >>low accrual,primary endpoint reached & investigator left NIH.) Collapse << | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02109250 | - | - | Completed | - | Belgium ... more >> Clinique du Sud Luxembourg Arlon, Belgium, 6700 AZ Klina Brasschaat, Belgium, 2930 Institut Jules Bordet Brussels, Belgium, 1000 UZ Brussel Brussels, Belgium, 1090 UCL St-Luc Brussels, Belgium, 1200 UZ Gent Gent, Belgium, 9000 AZ Delta Roeselare, Belgium, 8800 CH de Wallonie Tournai, Belgium, 7500 Collapse << |
| NCT00821080 | Glioblastoma | Phase 1 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Dana-Farber Cancer Instiute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01582191 | Advanced Cancers | Phase 1 | Recruiting | May 2026 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Collapse << |
| NCT01934335 | Invasive Breast Cancer | Phase 2 | Suspended(Drugs unavailable) | July 2019 | United States, Iowa ... more >> University of Iowa Hospitals and Clinics Iowa City, Iowa, United States, 52242 Collapse << |
| NCT01876784 | - | - | Active, not recruiting | - | - |
| NCT01876784 | Differentiated Thyroid Cancer | Phase 3 | Active, not recruiting | December 31, 2019 | - |
| NCT01298323 | Locally Advanced or Metastatic... more >> Medullary Thyroid Cancer Medullary Thyroid Cancer Collapse << | Phase 3 | Active, not recruiting | December 31, 2019 | - |
| NCT01496313 | - | - | Active, not recruiting | - | - |
| NCT00613223 | Gliosarcoma G... more >>lioblastoma Collapse << | Phase 1 | Completed | - | United States, North Carolina ... more >> Duke University Health System Durham, North Carolina, United States, 27710 Collapse << |
| NCT01298323 | - | - | Active, not recruiting | - | - |
| NCT01661179 | Unresectable Locally Advanced ... more >>or Metastatic, Medullary Thyroid Carcinoma Collapse << | Phase 1 Phase 2 | Completed | - | Japan ... more >> Research Site Fukuoka-shi, Japan Research Site Kobe-shi, Japan Research Site Koto-ku, Japan Research Site Shinjuku-ku, Japan Collapse << |
| NCT01661179 | - | - | Completed | - | - |
| NCT01539655 | Medullary Thyroid Cancer | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT01551615 | Healthy | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT00566995 | Renal Cancer ... more >>Von Hippel Lindau Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01823068 | Non Small Cell Lung Cancer | Phase 2 | Unknown | July 2018 | Korea, Republic of ... more >> Seoul National University Hospital Recruiting Seoul, Korea, Republic of, 110744 Principal Investigator: Se-Hoon Lee, M.D., Ph.D. Samsung Medical Center Recruiting Seoul, Korea, Republic of, 135710 Principal Investigator: Myung-Ju Ahn, M.D. Collapse << |
| NCT01561781 | Healthy Volunteers | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT03291379 | Carcinoma, Hepatocellular ... more >> Metastatic Colorectal Cancer Collapse << | Early Phase 1 | Recruiting | January 2019 | United Kingdom ... more >> University College London Hospital Recruiting Bloomsbury, London, United Kingdom, NW1 2BU Contact: Professor Ricky Sharma Collapse << |
| NCT00566995 | - | - | Completed | - | - |
| NCT01544140 | Healthy Volunteers | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| VEGFR2 | IC50:40nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
