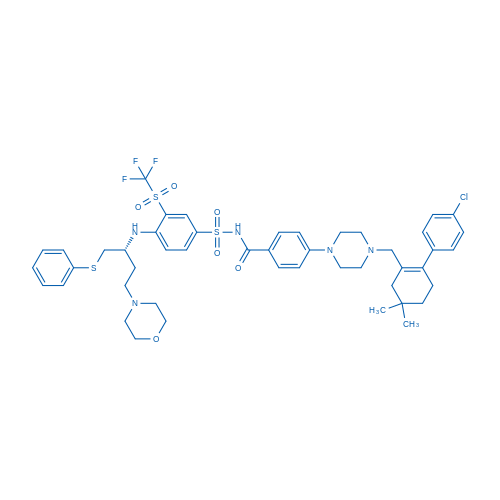
CAS No.: 923564-51-6
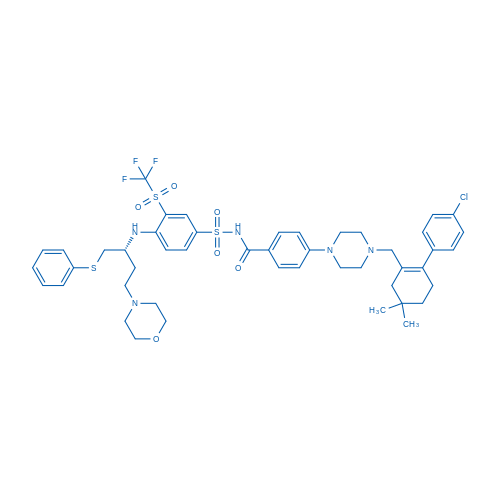
Navitoclax Catalog No. CSN12932
Synonyms: ABT-263;A-855071
ABT-263 is a potent inhibitor of Bcl-xL, Bcl-2 and Bcl-w with Ki of ≤ 0.5 nM, ≤1 nM and ≤1 nM in cell-free assays, but binds more weakly to Mcl-1 and A1.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 Bcl-2
Ki:1nMBcl-w
Ki:1nMBcl-xL
Ki:0.5nM- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01423539 Diffuse Large B-Cell Lymphoma Phase 2 Withdrawn(The NAVIGATE study h... more >>as been terminated due to non-safety related reasons.) Collapse << February 2014 United States, California ... more >> Fountain Valley, California, United States, 92708 United States, Florida Hudson, Florida, United States, 34667 United States, Georgia Lawrenceville, Georgia, United States, 30045 United States, Illinois Centralia, Illinois, United States, 62801 Harvey, Illinois, United States, 60426 United States, Indiana Terre Haute, Indiana, United States, 47802 United States, Kentucky Hazard, Kentucky, United States, 41701 Paducah, Kentucky, United States, 42003 United States, Maryland Rockville, Maryland, United States, 20850 United States, Missouri Jefferson City, Missouri, United States, 65109 United States, Montana Great Falls, Montana, United States, 59405 United States, Ohio Newark, Ohio, United States, 43055 United States, Tennessee Nashville, Tennessee, United States, 37203 Collapse << NCT01053520 Healthy Female Subjects Phase 1 Completed - United States, Illinois ... more >> Site Reference ID/Investigator# 23602 Waukegan, Illinois, United States, 60085 Collapse << NCT01989585 BRAF V600E Mutation Present ... more >> BRAF V600K Mutation Present Metastatic Melanoma Solid Neoplasm Stage III Cutaneous Melanoma AJCC v7 Stage IIIA Cutaneous Melanoma AJCC v7 Stage IIIB Cutaneous Melanoma AJCC v7 Stage IIIC Cutaneous Melanoma AJCC v7 Stage IV Cutaneous Melanoma AJCC v6 and v7 Collapse << Phase 1 Phase 2 Recruiting - United States, California ... more >> University of California Davis Comprehensive Cancer Center Suspended Sacramento, California, United States, 95817 United States, Massachusetts Massachusetts General Hospital Cancer Center Withdrawn Boston, Massachusetts, United States, 02114 United States, Missouri Siteman Cancer Center at West County Hospital Suspended Creve Coeur, Missouri, United States, 63141 Washington University School of Medicine Suspended Saint Louis, Missouri, United States, 63110 Siteman Cancer Center-South County Suspended Saint Louis, Missouri, United States, 63129 Siteman Cancer Center at Christian Hospital Suspended Saint Louis, Missouri, United States, 63136 Siteman Cancer Center at Saint Peters Hospital Suspended Saint Peters, Missouri, United States, 63376 United States, New Jersey Rutgers Cancer Institute of New Jersey Recruiting New Brunswick, New Jersey, United States, 08903 Contact: Site Public Contact 732-235-8675 Principal Investigator: Janice M. Mehnert United States, North Carolina Duke University Medical Center Recruiting Durham, North Carolina, United States, 27710 Contact: Site Public Contact 888-275-3853 Principal Investigator: James L. Abbruzzese United States, Ohio Ohio State University Comprehensive Cancer Center Recruiting Columbus, Ohio, United States, 43210 Contact: Site Public Contact 800-293-5066 Jamesline@osumc.edu Principal Investigator: Kari L. Kendra United States, Texas M D Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Site Public Contact 877-312-3961 Principal Investigator: Michael A. Davies Collapse << - 更多
- 参考文献
- [1] Tse C, Shoemaker AR, et al. ABT-263: a potent and orally bioavailable Bcl-2 family inhibitor. Cancer Res. 2008 May 1;68(9):3421-8.
- [2] Levy MA, Claxton DF, et al. Therapeutic inhibition of BCL-2 and related family members. Expert Opin Investig Drugs. 2017 Mar;26(3):293-301.
- [3] Del Gaizo Moore V, Brown JR, et al. Chronic lymphocytic leukemia requires BCL2 to sequester prodeath BIM, explaining sensitivity to BCL2 antagonist ABT-737. J Clin Invest. 2007 Jan;117(1):112-21.
- [4] Chen Q, Song S, et al. ABT-263 induces apoptosis and synergizes with chemotherapy by targeting stemness pathways in esophageal cancer. Oncotarget. 2015 Sep 22;6(28):25883-96.
- [5] Ackler S, Xiao Y, et al. ABT-263 and rapamycin act cooperatively to kill lymphoma cells in vitro and in vivo. Mol Cancer Ther. 2008 Oct;7(10):3265-74.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 923564-51-6 | 储存条件 |
|
|||||||||||||
| 分子式 | C47H55ClF3N5O6S3 | 运输 | 蓝冰 | |||||||||||||
| 分子量 | 974.61 | 别名 | ABT-263;A-855071 | |||||||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| 5637 | - | Growth Inhibition Assay | - | IC50=3.61103 μM | SANGER |
| 697 | - | Growth Inhibition Assay | - | IC50=0.10839 μM | SANGER |
| 769-P | - | Growth Inhibition Assay | - | IC50=3.8951 μM | SANGER |
| 8305C | - | Growth Inhibition Assay | - | IC50=35.8415 μM | SANGER |
| 8505C | - | Growth Inhibition Assay | - | IC50=4.5423 μM | SANGER |
| A204 | - | Growth Inhibition Assay | - | IC50=42.6388 μM | SANGER |
| A2058 | - | Growth Inhibition Assay | - | IC50=12.8681 μM | SANGER |
| A375 | - | Growth Inhibition Assay | - | IC50=30.3243 μM | SANGER |
| A388 | - | Growth Inhibition Assay | - | IC50=1.68724 μM | SANGER |
| A427 | - | Growth Inhibition Assay | - | IC50=0.93022 μM | SANGER |
| A431 | - | Growth Inhibition Assay | - | IC50=18.4127 μM | SANGER |
| A704 | - | Growth Inhibition Assay | - | IC50=0.4267 μM | SANGER |
| ABC-1 | - | Growth Inhibition Assay | - | IC50=17.8141 μM | SANGER |
| ACHN | - | Growth Inhibition Assay | - | IC50=11.4211 μM | SANGER |
| ACN | - | Growth Inhibition Assay | - | IC50=40.4911 μM | SANGER |
| AGS | - | Growth Inhibition Assay | - | IC50=10.374 μM | SANGER |
| ALL-PO | - | Growth Inhibition Assay | - | IC50=0.21988 μM | SANGER |
| AM-38 | - | Growth Inhibition Assay | - | IC50=13.2566 μM | SANGER |
| AN3-CA | - | Growth Inhibition Assay | - | IC50=3.44238 μM | SANGER |
| AsPC-1 | - | Growth Inhibition Assay | - | IC50=19.8657 μM | SANGER |
| ATN-1 | - | Growth Inhibition Assay | - | IC50=0.04733 μM | SANGER |
| BALL-1 | - | Growth Inhibition Assay | - | IC50=47.6168 μM | SANGER |
| BB30-HNC | - | Growth Inhibition Assay | - | IC50=21.2745 μM | SANGER |
| BB65-RCC | - | Growth Inhibition Assay | - | IC50=4.04399 μM | SANGER |
| BCPAP | - | Growth Inhibition Assay | - | IC50=4.63481 μM | SANGER |
| BE-13 | - | Growth Inhibition Assay | - | IC50=0.36459 μM | SANGER |
| BEN | - | Growth Inhibition Assay | - | IC50=0.23968 μM | SANGER |
| BHT-101 | - | Growth Inhibition Assay | - | IC50=10.7177 μM | SANGER |
| BHY | - | Growth Inhibition Assay | - | IC50=18.869 μM | SANGER |
| BPH-1 | - | Growth Inhibition Assay | - | IC50=18.1685 μM | SANGER |
| BT-20 | - | Growth Inhibition Assay | - | IC50=8.26052 μM | SANGER |
| BV-173 | - | Growth Inhibition Assay | - | IC50=0.02314 μM | SANGER |
| C2BBe1 | - | Growth Inhibition Assay | - | IC50=4.74008 μM | SANGER |
| C32 | - | Growth Inhibition Assay | - | IC50=1.19088 μM | SANGER |
| C8166 | - | Growth Inhibition Assay | - | IC50=28.6938 μM | SANGER |
| Ca9-22 | - | Growth Inhibition Assay | - | IC50=12.9451 μM | SANGER |
| CAL-120 | - | Growth Inhibition Assay | - | IC50=1.16429 μM | SANGER |
| CAL-12T | - | Growth Inhibition Assay | - | IC50=39.5281 μM | SANGER |
| CAL-27 | - | Growth Inhibition Assay | - | IC50=19.0487 μM | SANGER |
| CAL-54 | - | Growth Inhibition Assay | - | IC50=31.7241 μM | SANGER |
| Calu-3 | - | Growth Inhibition Assay | - | IC50=1.70697 μM | SANGER |
| Calu-6 | - | Growth Inhibition Assay | - | IC50=4.27881 μM | SANGER |
| CAMA-1 | - | Growth Inhibition Assay | - | IC50=20.9803 μM | SANGER |
| CAPAN-1 | - | Growth Inhibition Assay | - | IC50=7.77357 μM | SANGER |
| Capan-2 | - | Growth Inhibition Assay | - | IC50=18.9078 μM | SANGER |
| Ca-Ski | - | Growth Inhibition Assay | - | IC50=7.99383 μM | SANGER |
| CCRF-CEM | - | Growth Inhibition Assay | - | IC50=0.33529 μM | SANGER |
| CFPAC-1 | - | Growth Inhibition Assay | - | IC50=33.2332 μM | SANGER |
| CGTH-W-1 | - | Growth Inhibition Assay | - | IC50=17.5219 μM | SANGER |
| CHP-134 | - | Growth Inhibition Assay | - | IC50=0.16306 μM | SANGER |
| CHP-212 | - | Growth Inhibition Assay | - | IC50=3.92549 μM | SANGER |
| COLO-320-HSR | - | Growth Inhibition Assay | - | IC50=7.22738 μM | SANGER |
| COLO-668 | - | Growth Inhibition Assay | - | IC50=6.84387 μM | SANGER |
| COLO-680N | - | Growth Inhibition Assay | - | IC50=25.6329 μM | SANGER |
| COLO-684 | - | Growth Inhibition Assay | - | IC50=8.01818 μM | SANGER |
| COLO-792 | - | Growth Inhibition Assay | - | IC50=1.20071 μM | SANGER |
| COLO-800 | - | Growth Inhibition Assay | - | IC50=39.3868 μM | SANGER |
| COLO-824 | - | Growth Inhibition Assay | - | IC50=1.01708 μM | SANGER |
| COR-L105 | - | Growth Inhibition Assay | - | IC50=16.6528 μM | SANGER |
| COR-L23 | - | Growth Inhibition Assay | - | IC50=1.79874 μM | SANGER |
| COR-L88 | - | Growth Inhibition Assay | - | IC50=0.3654 μM | SANGER |
| CP50-MEL-B | - | Growth Inhibition Assay | - | IC50=9.75782 μM | SANGER |
| CTB-1 | - | Growth Inhibition Assay | - | IC50=21.4755 μM | SANGER |
| CTV-1 | - | Growth Inhibition Assay | - | IC50=9.56532 μM | SANGER |
| D-247MG | - | Growth Inhibition Assay | - | IC50=1.51122 μM | SANGER |
| D-283MED | - | Growth Inhibition Assay | - | IC50=0.17686 μM | SANGER |
| D-566MG | - | Growth Inhibition Assay | - | IC50=41.2086 μM | SANGER |
| DB | - | Growth Inhibition Assay | - | IC50=0.5708 μM | SANGER |
| DEL | - | Growth Inhibition Assay | - | IC50=1.74692 μM | SANGER |
| DJM-1 | - | Growth Inhibition Assay | - | IC50=27.1454 μM | SANGER |
| DOHH-2 | - | Growth Inhibition Assay | - | IC50=0.41023 μM | SANGER |
| DoTc2-4510 | - | Growth Inhibition Assay | - | IC50=3.01417 μM | SANGER |
| DSH1 | - | Growth Inhibition Assay | - | IC50=45.0033 μM | SANGER |
| DU-145 | - | Growth Inhibition Assay | - | IC50=0.89923 μM | SANGER |
| DU-4475 | - | Growth Inhibition Assay | - | IC50=0.65427 μM | SANGER |
| ECC10 | - | Growth Inhibition Assay | - | IC50=0.03792 μM | SANGER |
| EFM-19 | - | Growth Inhibition Assay | - | IC50=3.48339 μM | SANGER |
| EGI-1 | - | Growth Inhibition Assay | - | IC50=42.428 μM | SANGER |
| EKVX | - | Growth Inhibition Assay | - | IC50=20.606 μM | SANGER |
| EM-2 | - | Growth Inhibition Assay | - | IC50=0.19918 μM | SANGER |
| EoL-1-cell | - | Growth Inhibition Assay | - | IC50=0.00669 μM | SANGER |
| EPLC-272H | - | Growth Inhibition Assay | - | IC50=9.37657 μM | SANGER |
| ES1 | - | Growth Inhibition Assay | - | IC50=32.3062 μM | SANGER |
| ES3 | - | Growth Inhibition Assay | - | IC50=4.35441 μM | SANGER |
| ES4 | - | Growth Inhibition Assay | - | IC50=1.34998 μM | SANGER |
| ES5 | - | Growth Inhibition Assay | - | IC50=4.13985 μM | SANGER |
| ES7 | - | Growth Inhibition Assay | - | IC50=1.27951 μM | SANGER |
| ES8 | - | Growth Inhibition Assay | - | IC50=2.25471 μM | SANGER |
| ESS-1 | - | Growth Inhibition Assay | - | IC50=5.25597 μM | SANGER |
| ETK-1 | - | Growth Inhibition Assay | - | IC50=39.022 μM | SANGER |
| EW-1 | - | Growth Inhibition Assay | - | IC50=15.5462 μM | SANGER |
| EW-11 | - | Growth Inhibition Assay | - | IC50=1.95657 μM | SANGER |
| EW-13 | - | Growth Inhibition Assay | - | IC50=0.58341 μM | SANGER |
| EW-16 | - | Growth Inhibition Assay | - | IC50=0.81328 μM | SANGER |
| EW-18 | - | Growth Inhibition Assay | - | IC50=0.06841 μM | SANGER |
| EW-22 | - | Growth Inhibition Assay | - | IC50=0.46671 μM | SANGER |
| EW-3 | - | Growth Inhibition Assay | - | IC50=0.08053 μM | SANGER |
| FTC-133 | - | Growth Inhibition Assay | - | IC50=4.5395 μM | SANGER |
| G-402 | - | Growth Inhibition Assay | - | IC50=5.51865 μM | SANGER |
| GAMG | - | Growth Inhibition Assay | - | IC50=10.5162 μM | SANGER |
| GCIY | - | Growth Inhibition Assay | - | IC50=3.20491 μM | SANGER |
| GCT | - | Growth Inhibition Assay | - | IC50=7.56906 μM | SANGER |
| GI-1 | - | Growth Inhibition Assay | - | IC50=5.27926 μM | SANGER |
| GMS-10 | - | Growth Inhibition Assay | - | IC50=26.8638 μM | SANGER |
| GOTO | - | Growth Inhibition Assay | - | IC50=2.65377 μM | SANGER |
| GP5d | - | Growth Inhibition Assay | - | IC50=4.78388 μM | SANGER |
| H9 | - | Growth Inhibition Assay | - | IC50=3.67144 μM | SANGER |
| HAL-01 | - | Growth Inhibition Assay | - | IC50=0.72549 μM | SANGER |
| HCC1395 | - | Growth Inhibition Assay | - | IC50=34.7186 μM | SANGER |
| HCC1419 | - | Growth Inhibition Assay | - | IC50=4.90063 μM | SANGER |
| HCC1569 | - | Growth Inhibition Assay | - | IC50=0.25106 μM | SANGER |
| HCC1806 | - | Growth Inhibition Assay | - | IC50=7.26044 μM | SANGER |
| HCC1937 | - | Growth Inhibition Assay | - | IC50=6.99251 μM | SANGER |
| HCC2157 | - | Growth Inhibition Assay | - | IC50=1.19494 μM | SANGER |
| HCC2218 | - | Growth Inhibition Assay | - | IC50=38.6519 μM | SANGER |
| HCC2998 | - | Growth Inhibition Assay | - | IC50=1.07135 μM | SANGER |
| HCC38 | - | Growth Inhibition Assay | - | IC50=0.8019 μM | SANGER |
| HCC70 | - | Growth Inhibition Assay | - | IC50=32.1387 μM | SANGER |
| HCE-T | - | Growth Inhibition Assay | - | IC50=7.87783 μM | SANGER |
| HDLM-2 | - | Growth Inhibition Assay | - | IC50=33.6931 μM | SANGER |
| HD-MY-Z | - | Growth Inhibition Assay | - | IC50=5.23969 μM | SANGER |
| HEC-1 | - | Growth Inhibition Assay | - | IC50=10.3352 μM | SANGER |
| HEL | - | Growth Inhibition Assay | - | IC50=1.31029 μM | SANGER |
| H-EMC-SS | - | Growth Inhibition Assay | - | IC50=4.31831 μM | SANGER |
| HH | - | Growth Inhibition Assay | - | IC50=3.83868 μM | SANGER |
| HL-60 | - | Growth Inhibition Assay | - | IC50=0.02908 μM | SANGER |
| HLE | - | Growth Inhibition Assay | - | IC50=44.0856 μM | SANGER |
| HMV-II | - | Growth Inhibition Assay | - | IC50=31.3592 μM | SANGER |
| HN | - | Growth Inhibition Assay | - | IC50=15.5942 μM | SANGER |
| HO-1-N-1 | - | Growth Inhibition Assay | - | IC50=48.7445 μM | SANGER |
| HOP-92 | - | Growth Inhibition Assay | - | IC50=2.58743 μM | SANGER |
| HPAF-II | - | Growth Inhibition Assay | - | IC50=0.92628 μM | SANGER |
| HSC-2 | - | Growth Inhibition Assay | - | IC50=9.5755 μM | SANGER |
| HSC-3 | - | Growth Inhibition Assay | - | IC50=1.96365 μM | SANGER |
| HT-1080 | - | Growth Inhibition Assay | - | IC50=9.77739 μM | SANGER |
| HT-3 | - | Growth Inhibition Assay | - | IC50=17.5914 μM | SANGER |
| HTC-C3 | - | Growth Inhibition Assay | - | IC50=22.5555 μM | SANGER |
| HuH-7 | - | Growth Inhibition Assay | - | IC50=1.67293 μM | SANGER |
| HuO-3N1 | - | Growth Inhibition Assay | - | IC50=2.4878 μM | SANGER |
| HuO9 | - | Growth Inhibition Assay | - | IC50=0.26715 μM | SANGER |
| HuP-T4 | - | Growth Inhibition Assay | - | IC50=17.2495 μM | SANGER |
| IA-LM | - | Growth Inhibition Assay | - | IC50=5.39871 μM | SANGER |
| IGR-1 | - | Growth Inhibition Assay | - | IC50=2.62035 μM | SANGER |
| IST-MEL1 | - | Growth Inhibition Assay | - | IC50=21.3692 μM | SANGER |
| IST-SL1 | - | Growth Inhibition Assay | - | IC50=0.35343 μM | SANGER |
| J82 | - | Growth Inhibition Assay | - | IC50=8.90038 μM | SANGER |
| J-RT3-T3-5 | - | Growth Inhibition Assay | - | IC50=0.60808 μM | SANGER |
| JVM-3 | - | Growth Inhibition Assay | - | IC50=4.18222 μM | SANGER |
| K5 | - | Growth Inhibition Assay | - | IC50=45.9405 μM | SANGER |
| KALS-1 | - | Growth Inhibition Assay | - | IC50=48.1289 μM | SANGER |
| KARPAS-299 | - | Growth Inhibition Assay | - | IC50=3.67361 μM | SANGER |
| KARPAS-45 | - | Growth Inhibition Assay | - | IC50=0.07815 μM | SANGER |
| KE-37 | - | Growth Inhibition Assay | - | IC50=0.1137 μM | SANGER |
| KG-1 | - | Growth Inhibition Assay | - | IC50=0.04486 μM | SANGER |
| KINGS-1 | - | Growth Inhibition Assay | - | IC50=5.07744 μM | SANGER |
| KM12 | - | Growth Inhibition Assay | - | IC50=36.7547 μM | SANGER |
| KNS-42 | - | Growth Inhibition Assay | - | IC50=12.9984 μM | SANGER |
| KNS-62 | - | Growth Inhibition Assay | - | IC50=16.9777 μM | SANGER |
| KNS-81-FD | - | Growth Inhibition Assay | - | IC50=0.44017 μM | SANGER |
| KP-4 | - | Growth Inhibition Assay | - | IC50=5.46334 μM | SANGER |
| KP-N-YN | - | Growth Inhibition Assay | - | IC50=0.23573 μM | SANGER |
| KP-N-YS | - | Growth Inhibition Assay | - | IC50=1.16236 μM | SANGER |
| KS-1 | - | Growth Inhibition Assay | - | IC50=17.673 μM | SANGER |
| KU812 | - | Growth Inhibition Assay | - | IC50=19.9573 μM | SANGER |
| KY821 | - | Growth Inhibition Assay | - | IC50=0.02975 μM | SANGER |
| KYSE-410 | - | Growth Inhibition Assay | - | IC50=16.591 μM | SANGER |
| KYSE-520 | - | Growth Inhibition Assay | - | IC50=25.644 μM | SANGER |
| KYSE-70 | - | Growth Inhibition Assay | - | IC50=8.07729 μM | SANGER |
| L-363 | - | Growth Inhibition Assay | - | IC50=13.4033 μM | SANGER |
| L-428 | - | Growth Inhibition Assay | - | IC50=1.0125 μM | SANGER |
| LAMA-84 | - | Growth Inhibition Assay | - | IC50=0.59207 μM | SANGER |
| LAN-6 | - | Growth Inhibition Assay | - | IC50=4.45189 μM | SANGER |
| LB1047-RCC | - | Growth Inhibition Assay | - | IC50=6.47625 μM | SANGER |
| LB2241-RCC | - | Growth Inhibition Assay | - | IC50=9.19137 μM | SANGER |
| LB2518-MEL | - | Growth Inhibition Assay | - | IC50=16.8389 μM | SANGER |
| LB831-BLC | - | Growth Inhibition Assay | - | IC50=15.2767 μM | SANGER |
| LB996-RCC | - | Growth Inhibition Assay | - | IC50=9.1989 μM | SANGER |
| LC-2-ad | - | Growth Inhibition Assay | - | IC50=4.29568 μM | SANGER |
| LCLC-103H | - | Growth Inhibition Assay | - | IC50=22.1582 μM | SANGER |
| LK-2 | - | Growth Inhibition Assay | - | IC50=4.11293 μM | SANGER |
| LoVo | - | Growth Inhibition Assay | - | IC50=13.2313 μM | SANGER |
| LU-134-A | - | Growth Inhibition Assay | - | IC50=0.18671 μM | SANGER |
| LU-135 | - | Growth Inhibition Assay | - | IC50=0.18552 μM | SANGER |
| LU-139 | - | Growth Inhibition Assay | - | IC50=0.20498 μM | SANGER |
| LXF-289 | - | Growth Inhibition Assay | - | IC50=5.03719 μM | SANGER |
| M059J | - | Growth Inhibition Assay | - | IC50=12.7727 μM | SANGER |
| MC116 | - | Growth Inhibition Assay | - | IC50=6.93897 μM | SANGER |
| MCF7 | - | Growth Inhibition Assay | - | IC50=45.5051 μM | SANGER |
| MC-IXC | - | Growth Inhibition Assay | - | IC50=12.7518 μM | SANGER |
| MDA-MB-157 | - | Growth Inhibition Assay | - | IC50=4.8876 μM | SANGER |
| MDA-MB-175-VII | - | Growth Inhibition Assay | - | IC50=18.9209 μM | SANGER |
| MDA-MB-415 | - | Growth Inhibition Assay | - | IC50=1.3885 μM | SANGER |
| ME-180 | - | Growth Inhibition Assay | - | IC50=12.7054 μM | SANGER |
| MEG-01 | - | Growth Inhibition Assay | - | IC50=0.5832 μM | SANGER |
| MEL-HO | - | Growth Inhibition Assay | - | IC50=7.77054 μM | SANGER |
| Mewo | - | Growth Inhibition Assay | - | IC50=20.1288 μM | SANGER |
| MFE-280 | - | Growth Inhibition Assay | - | IC50=2.02848 μM | SANGER |
| MFM-223 | - | Growth Inhibition Assay | - | IC50=15.1813 μM | SANGER |
| MHH-ES-1 | - | Growth Inhibition Assay | - | IC50=8.51834 μM | SANGER |
| MHH-NB-11 | - | Growth Inhibition Assay | - | IC50=2.51158 μM | SANGER |
| MKN28 | - | Growth Inhibition Assay | - | IC50=33.3809 μM | SANGER |
| MKN7 | - | Growth Inhibition Assay | - | IC50=15.9732 μM | SANGER |
| ML-2 | - | Growth Inhibition Assay | - | IC50=0.01983 μM | SANGER |
| MLMA | - | Growth Inhibition Assay | - | IC50=1.96677 μM | SANGER |
| MMAC-SF | - | Growth Inhibition Assay | - | IC50=9.6875 μM | SANGER |
| MN-60 | - | Growth Inhibition Assay | - | IC50=11.539 μM | SANGER |
| MOLT-13 | - | Growth Inhibition Assay | - | IC50=0.46613 μM | SANGER |
| MOLT-16 | - | Growth Inhibition Assay | - | IC50=0.65264 μM | SANGER |
| MOLT-4 | - | Growth Inhibition Assay | - | IC50=0.15169 μM | SANGER |
| Mo-T | - | Growth Inhibition Assay | - | IC50=4.04312 μM | SANGER |
| MPP-89 | - | Growth Inhibition Assay | - | IC50=3.05666 μM | SANGER |
| MS-1 | - | Growth Inhibition Assay | - | IC50=0.46933 μM | SANGER |
| MV-4-11 | - | Growth Inhibition Assay | - | IC50=0.01586 μM | SANGER |
| NB10 | - | Growth Inhibition Assay | - | IC50=26.3117 μM | SANGER |
| NB12 | - | Growth Inhibition Assay | - | IC50=0.23115 μM | SANGER |
| NB13 | - | Growth Inhibition Assay | - | IC50=0.08203 μM | SANGER |
| NB14 | - | Growth Inhibition Assay | - | IC50=1.13748 μM | SANGER |
| NB17 | - | Growth Inhibition Assay | - | IC50=0.07124 μM | SANGER |
| NB5 | - | Growth Inhibition Assay | - | IC50=13.3752 μM | SANGER |
| NB6 | - | Growth Inhibition Assay | - | IC50=4.86204 μM | SANGER |
| NB69 | - | Growth Inhibition Assay | - | IC50=1.46271 μM | SANGER |
| NBsusSR | - | Growth Inhibition Assay | - | IC50=3.34938 μM | SANGER |
| NCI-H1048 | - | Growth Inhibition Assay | - | IC50=0.50953 μM | SANGER |
| NCI-H1092 | - | Growth Inhibition Assay | - | IC50=0.10275 μM | SANGER |
| NCI-H1155 | - | Growth Inhibition Assay | - | IC50=5.54955 μM | SANGER |
| NCI-H1304 | - | Growth Inhibition Assay | - | IC50=0.46157 μM | SANGER |
| NCI-H1355 | - | Growth Inhibition Assay | - | IC50=33.204 μM | SANGER |
| NCI-H1563 | - | Growth Inhibition Assay | - | IC50=14.6039 μM | SANGER |
| NCI-H1581 | - | Growth Inhibition Assay | - | IC50=2.79681 μM | SANGER |
| NCI-H1623 | - | Growth Inhibition Assay | - | IC50=4.70228 μM | SANGER |
| NCI-H1648 | - | Growth Inhibition Assay | - | IC50=1.71418 μM | SANGER |
| NCI-H1650 | - | Growth Inhibition Assay | - | IC50=2.72215 μM | SANGER |
| NCI-H1693 | - | Growth Inhibition Assay | - | IC50=28.6975 μM | SANGER |
| NCI-H1770 | - | Growth Inhibition Assay | - | IC50=6.20874 μM | SANGER |
| NCI-H1792 | - | Growth Inhibition Assay | - | IC50=17.798 μM | SANGER |
| NCI-H2029 | - | Growth Inhibition Assay | - | IC50=4.94185 μM | SANGER |
| NCI-H2030 | - | Growth Inhibition Assay | - | IC50=47.2374 μM | SANGER |
| NCI-H2052 | - | Growth Inhibition Assay | - | IC50=1.72201 μM | SANGER |
| NCI-H2087 | - | Growth Inhibition Assay | - | IC50=31.6352 μM | SANGER |
| NCI-H209 | - | Growth Inhibition Assay | - | IC50=0.08704 μM | SANGER |
| NCI-H2122 | - | Growth Inhibition Assay | - | IC50=26.9998 μM | SANGER |
| NCI-H2126 | - | Growth Inhibition Assay | - | IC50=7.73625 μM | SANGER |
| NCI-H2170 | - | Growth Inhibition Assay | - | IC50=1.0623 μM | SANGER |
| NCI-H2228 | - | Growth Inhibition Assay | - | IC50=14.6077 μM | SANGER |
| NCI-H2291 | - | Growth Inhibition Assay | - | IC50=11.5466 μM | SANGER |
| NCI-H23 | - | Growth Inhibition Assay | - | IC50=21.1277 μM | SANGER |
| NCI-H2342 | - | Growth Inhibition Assay | - | IC50=1.40269 μM | SANGER |
| NCI-H2347 | - | Growth Inhibition Assay | - | IC50=12.7614 μM | SANGER |
| NCI-H2405 | - | Growth Inhibition Assay | - | IC50=2.82782 μM | SANGER |
| NCI-H2452 | - | Growth Inhibition Assay | - | IC50=49.1152 μM | SANGER |
| NCI-H292 | - | Growth Inhibition Assay | - | IC50=25.3536 μM | SANGER |
| NCI-H358 | - | Growth Inhibition Assay | - | IC50=47.215 μM | SANGER |
| NCI-H441 | - | Growth Inhibition Assay | - | IC50=20.001 μM | SANGER |
| NCI-H446 | - | Growth Inhibition Assay | - | IC50=6.62925 μM | SANGER |
| NCI-H522 | - | Growth Inhibition Assay | - | IC50=15.3306 μM | SANGER |
| NCI-H526 | - | Growth Inhibition Assay | - | IC50=11.5043 μM | SANGER |
| NCI-H596 | - | Growth Inhibition Assay | - | IC50=3.04997 μM | SANGER |
| NCI-H630 | - | Growth Inhibition Assay | - | IC50=5.99404 μM | SANGER |
| NCI-H661 | - | Growth Inhibition Assay | - | IC50=26.4713 μM | SANGER |
| NCI-H69 | - | Growth Inhibition Assay | - | IC50=12.4234 μM | SANGER |
| NCI-H720 | - | Growth Inhibition Assay | - | IC50=0.04011 μM | SANGER |
| NCI-H727 | - | Growth Inhibition Assay | - | IC50=20.2704 μM | SANGER |
| NCI-H82 | - | Growth Inhibition Assay | - | IC50=3.95936 μM | SANGER |
| NCI-N87 | - | Growth Inhibition Assay | - | IC50=7.19293 μM | SANGER |
| NCI-SNU-1 | - | Growth Inhibition Assay | - | IC50=1.36555 μM | SANGER |
| NEC8 | - | Growth Inhibition Assay | - | IC50=6.7663 μM | SANGER |
| NH-12 | - | Growth Inhibition Assay | - | IC50=0.10744 μM | SANGER |
| NKM-1 | - | Growth Inhibition Assay | - | IC50=0.01699 μM | SANGER |
| NMC-G1 | - | Growth Inhibition Assay | - | IC50=0.81121 μM | SANGER |
| no-11 | - | Growth Inhibition Assay | - | IC50=4.35554 μM | SANGER |
| NOMO-1 | - | Growth Inhibition Assay | - | IC50=6.78111 μM | SANGER |
| NOS-1 | - | Growth Inhibition Assay | - | IC50=3.79834 μM | SANGER |
| NTERA-S-cl-D1 | - | Growth Inhibition Assay | - | IC50=0.50019 μM | SANGER |
| NUGC-3 | - | Growth Inhibition Assay | - | IC50=7.39694 μM | SANGER |
| OAW-28 | - | Growth Inhibition Assay | - | IC50=0.7837 μM | SANGER |
| OAW-42 | - | Growth Inhibition Assay | - | IC50=0.96146 μM | SANGER |
| OC-314 | - | Growth Inhibition Assay | - | IC50=49.6834 μM | SANGER |
| OCI-AML2 | - | Growth Inhibition Assay | - | IC50=3.31272 μM | SANGER |
| OE19 | - | Growth Inhibition Assay | - | IC50=5.68624 μM | SANGER |
| OMC-1 | - | Growth Inhibition Assay | - | IC50=1.86016 μM | SANGER |
| OS-RC-2 | - | Growth Inhibition Assay | - | IC50=5.52604 μM | SANGER |
| OVCAR-3 | - | Growth Inhibition Assay | - | IC50=7.33038 μM | SANGER |
| OVCAR-4 | - | Growth Inhibition Assay | - | IC50=16.8862 μM | SANGER |
| OVCAR-5 | - | Growth Inhibition Assay | - | IC50=10.6429 μM | SANGER |
| OVCAR-8 | - | Growth Inhibition Assay | - | IC50=27.0638 μM | SANGER |
| P12-ICHIKAWA | - | Growth Inhibition Assay | - | IC50=0.07778 μM | SANGER |
| P30-OHK | - | Growth Inhibition Assay | - | IC50=1.04688 μM | SANGER |
| PA-1 | - | Growth Inhibition Assay | - | IC50=0.95642 μM | SANGER |
| PANC-03-27 | - | Growth Inhibition Assay | - | IC50=22.5169 μM | SANGER |
| PANC-08-13 | - | Growth Inhibition Assay | - | IC50=3.63472 μM | SANGER |
| PANC-10-05 | - | Growth Inhibition Assay | - | IC50=34.1014 μM | SANGER |
| PFSK-1 | - | Growth Inhibition Assay | - | IC50=6.05259 μM | SANGER |
| PSN1 | - | Growth Inhibition Assay | - | IC50=41.1749 μM | SANGER |
| QIMR-WIL | - | Growth Inhibition Assay | - | IC50=0.04287 μM | SANGER |
| Ramos-2G6-4C10 | - | Growth Inhibition Assay | - | IC50=1.73656 μM | SANGER |
| RCC10RGB | - | Growth Inhibition Assay | - | IC50=8.99561 μM | SANGER |
| RCM-1 | - | Growth Inhibition Assay | - | IC50=2.00399 μM | SANGER |
| RD | - | Growth Inhibition Assay | - | IC50=0.75899 μM | SANGER |
| RH-18 | - | Growth Inhibition Assay | - | IC50=0.06048 μM | SANGER |
| RMG-I | - | Growth Inhibition Assay | - | IC50=0.49464 μM | SANGER |
| RPMI-2650 | - | Growth Inhibition Assay | - | IC50=5.3616 μM | SANGER |
| RPMI-7951 | - | Growth Inhibition Assay | - | IC50=4.22413 μM | SANGER |
| RPMI-8226 | - | Growth Inhibition Assay | - | IC50=0.45652 μM | SANGER |
| RPMI-8866 | - | Growth Inhibition Assay | - | IC50=17.1732 μM | SANGER |
| RS4-11 | - | Growth Inhibition Assay | - | IC50=0.02587 μM | SANGER |
| RT-112 | - | Growth Inhibition Assay | - | IC50=20.6122 μM | SANGER |
| RVH-421 | - | Growth Inhibition Assay | - | IC50=3.56877 μM | SANGER |
| Saos-2 | - | Growth Inhibition Assay | - | IC50=42.8369 μM | SANGER |
| SAS | - | Growth Inhibition Assay | - | IC50=34.4565 μM | SANGER |
| SBC-1 | - | Growth Inhibition Assay | - | IC50=1.19053 μM | SANGER |
| SCC-15 | - | Growth Inhibition Assay | - | IC50=5.93662 μM | SANGER |
| SCC-25 | - | Growth Inhibition Assay | - | IC50=11.7556 μM | SANGER |
| SCC-4 | - | Growth Inhibition Assay | - | IC50=1.59887 μM | SANGER |
| SCC-9 | - | Growth Inhibition Assay | - | IC50=15.8114 μM | SANGER |
| SF126 | - | Growth Inhibition Assay | - | IC50=8.83865 μM | SANGER |
| SF295 | - | Growth Inhibition Assay | - | IC50=10.6704 μM | SANGER |
| SJSA-1 | - | Growth Inhibition Assay | - | IC50=41.1596 μM | SANGER |
| SKG-IIIa | - | Growth Inhibition Assay | - | IC50=7.81892 μM | SANGER |
| SK-LU-1 | - | Growth Inhibition Assay | - | IC50=4.66089 μM | SANGER |
| SK-MEL-1 | - | Growth Inhibition Assay | - | IC50=38.3389 μM | SANGER |
| SK-MEL-2 | - | Growth Inhibition Assay | - | IC50=11.7637 μM | SANGER |
| SK-MEL-24 | - | Growth Inhibition Assay | - | IC50=20.1477 μM | SANGER |
| SK-MEL-28 | - | Growth Inhibition Assay | - | IC50=9.61893 μM | SANGER |
| SK-MEL-3 | - | Growth Inhibition Assay | - | IC50=6.42915 μM | SANGER |
| SK-MEL-30 | - | Growth Inhibition Assay | - | IC50=14.0645 μM | SANGER |
| SK-N-DZ | - | Growth Inhibition Assay | - | IC50=16.6116 μM | SANGER |
| SK-NEP-1 | - | Growth Inhibition Assay | - | IC50=0.07213 μM | SANGER |
| SK-N-FI | - | Growth Inhibition Assay | - | IC50=48.0358 μM | SANGER |
| SK-PN-DW | - | Growth Inhibition Assay | - | IC50=21.1649 μM | SANGER |
| SK-UT-1 | - | Growth Inhibition Assay | - | IC50=9.04945 μM | SANGER |
| SN12C | - | Growth Inhibition Assay | - | IC50=11.9355 μM | SANGER |
| SNU-423 | - | Growth Inhibition Assay | - | IC50=1.78157 μM | SANGER |
| SNU-C2B | - | Growth Inhibition Assay | - | IC50=43.6878 μM | SANGER |
| SW1116 | - | Growth Inhibition Assay | - | IC50=37.5992 μM | SANGER |
| SW1417 | - | Growth Inhibition Assay | - | IC50=0.55438 μM | SANGER |
| SW1463 | - | Growth Inhibition Assay | - | IC50=44.9971 μM | SANGER |
| SW1710 | - | Growth Inhibition Assay | - | IC50=30.126 μM | SANGER |
| SW1783 | - | Growth Inhibition Assay | - | IC50=7.43175 μM | SANGER |
| SW1990 | - | Growth Inhibition Assay | - | IC50=4.05908 μM | SANGER |
| SW48 | - | Growth Inhibition Assay | - | IC50=10.5189 μM | SANGER |
| SW620 | - | Growth Inhibition Assay | - | IC50=4.57057 μM | SANGER |
| SW626 | - | Growth Inhibition Assay | - | IC50=3.24543 μM | SANGER |
| SW756 | - | Growth Inhibition Assay | - | IC50=7.78333 μM | SANGER |
| SW780 | - | Growth Inhibition Assay | - | IC50=6.70185 μM | SANGER |
| SW837 | - | Growth Inhibition Assay | - | IC50=15.7847 μM | SANGER |
| SW872 | - | Growth Inhibition Assay | - | IC50=15.3086 μM | SANGER |
| SW900 | - | Growth Inhibition Assay | - | IC50=21.0149 μM | SANGER |
| SW948 | - | Growth Inhibition Assay | - | IC50=47.464 μM | SANGER |
| SW954 | - | Growth Inhibition Assay | - | IC50=4.2966 μM | SANGER |
| T-24 | - | Growth Inhibition Assay | - | IC50=17.5347 μM | SANGER |
| T84 | - | Growth Inhibition Assay | - | IC50=38.7409 μM | SANGER |
| T98G | - | Growth Inhibition Assay | - | IC50=18.5157 μM | SANGER |
| TE-11 | - | Growth Inhibition Assay | - | IC50=2.29473 μM | SANGER |
| TE-12 | - | Growth Inhibition Assay | - | IC50=8.59931 μM | SANGER |
| TE-5 | - | Growth Inhibition Assay | - | IC50=3.70709 μM | SANGER |
| TE-8 | - | Growth Inhibition Assay | - | IC50=23.2565 μM | SANGER |
| TE-9 | - | Growth Inhibition Assay | - | IC50=47.9581 μM | SANGER |
| TGBC11TKB | - | Growth Inhibition Assay | - | IC50=2.57681 μM | SANGER |
| TGBC1TKB | - | Growth Inhibition Assay | - | IC50=1.14152 μM | SANGER |
| TGBC24TKB | - | Growth Inhibition Assay | - | IC50=0.45778 μM | SANGER |
| TGW | - | Growth Inhibition Assay | - | IC50=0.04633 μM | SANGER |
| TI-73 | - | Growth Inhibition Assay | - | IC50=8.25851 μM | SANGER |
| TK10 | - | Growth Inhibition Assay | - | IC50=16.9473 μM | SANGER |
| TYK-nu | - | Growth Inhibition Assay | - | IC50=30.0345 μM | SANGER |
| U031 | - | Growth Inhibition Assay | - | IC50=10.5908 μM | SANGER |
| U-118-MG | - | Growth Inhibition Assay | - | IC50=2.96491 μM | SANGER |
| U-2-OS | - | Growth Inhibition Assay | - | IC50=5.89013 μM | SANGER |
| UACC-893 | - | Growth Inhibition Assay | - | IC50=27.9878 μM | SANGER |
| UMC-11 | - | Growth Inhibition Assay | - | IC50=4.88964 μM | SANGER |
| VA-ES-BJ | - | Growth Inhibition Assay | - | IC50=12.8785 μM | SANGER |
| VM-CUB-1 | - | Growth Inhibition Assay | - | IC50=21.3536 μM | SANGER |
| VMRC-RCZ | - | Growth Inhibition Assay | - | IC50=11.3201 μM | SANGER |
| WM-115 | - | Growth Inhibition Assay | - | IC50=0.27738 μM | SANGER |
| YH-13 | - | Growth Inhibition Assay | - | IC50=8.61008 μM | SANGER |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT01423539 | Diffuse Large B-Cell Lymphoma | Phase 2 | Withdrawn(The NAVIGATE study h... more >>as been terminated due to non-safety related reasons.) Collapse << | February 2014 | United States, California ... more >> Fountain Valley, California, United States, 92708 United States, Florida Hudson, Florida, United States, 34667 United States, Georgia Lawrenceville, Georgia, United States, 30045 United States, Illinois Centralia, Illinois, United States, 62801 Harvey, Illinois, United States, 60426 United States, Indiana Terre Haute, Indiana, United States, 47802 United States, Kentucky Hazard, Kentucky, United States, 41701 Paducah, Kentucky, United States, 42003 United States, Maryland Rockville, Maryland, United States, 20850 United States, Missouri Jefferson City, Missouri, United States, 65109 United States, Montana Great Falls, Montana, United States, 59405 United States, Ohio Newark, Ohio, United States, 43055 United States, Tennessee Nashville, Tennessee, United States, 37203 Collapse << |
| NCT01053520 | Healthy Female Subjects | Phase 1 | Completed | - | United States, Illinois ... more >> Site Reference ID/Investigator# 23602 Waukegan, Illinois, United States, 60085 Collapse << |
| NCT01989585 | BRAF V600E Mutation Present ... more >> BRAF V600K Mutation Present Metastatic Melanoma Solid Neoplasm Stage III Cutaneous Melanoma AJCC v7 Stage IIIA Cutaneous Melanoma AJCC v7 Stage IIIB Cutaneous Melanoma AJCC v7 Stage IIIC Cutaneous Melanoma AJCC v7 Stage IV Cutaneous Melanoma AJCC v6 and v7 Collapse << | Phase 1 Phase 2 | Recruiting | - | United States, California ... more >> University of California Davis Comprehensive Cancer Center Suspended Sacramento, California, United States, 95817 United States, Massachusetts Massachusetts General Hospital Cancer Center Withdrawn Boston, Massachusetts, United States, 02114 United States, Missouri Siteman Cancer Center at West County Hospital Suspended Creve Coeur, Missouri, United States, 63141 Washington University School of Medicine Suspended Saint Louis, Missouri, United States, 63110 Siteman Cancer Center-South County Suspended Saint Louis, Missouri, United States, 63129 Siteman Cancer Center at Christian Hospital Suspended Saint Louis, Missouri, United States, 63136 Siteman Cancer Center at Saint Peters Hospital Suspended Saint Peters, Missouri, United States, 63376 United States, New Jersey Rutgers Cancer Institute of New Jersey Recruiting New Brunswick, New Jersey, United States, 08903 Contact: Site Public Contact 732-235-8675 Principal Investigator: Janice M. Mehnert United States, North Carolina Duke University Medical Center Recruiting Durham, North Carolina, United States, 27710 Contact: Site Public Contact 888-275-3853 Principal Investigator: James L. Abbruzzese United States, Ohio Ohio State University Comprehensive Cancer Center Recruiting Columbus, Ohio, United States, 43210 Contact: Site Public Contact 800-293-5066 Jamesline@osumc.edu Principal Investigator: Kari L. Kendra United States, Texas M D Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Site Public Contact 877-312-3961 Principal Investigator: Michael A. Davies Collapse << |
| NCT02520778 | EGFR Activating Mutation ... more >> EGFR Exon 19 Deletion Mutation EGFR NP_005219.2:p.G719X EGFR NP_005219.2:p.L858R EGFR NP_005219.2:p.L861Q EGFR NP_005219.2:p.T790M Stage III Non-Small Cell Lung Cancer AJCC v7 Stage IIIA Non-Small Cell Lung Cancer AJCC v7 Stage IIIB Lung Non-Small Cell Cancer AJCC v7 Stage IV Non-Small Cell Lung Cancer AJCC v7 Collapse << | Phase 1 | Recruiting | - | United States, California ... more >> University of California Davis Comprehensive Cancer Center Recruiting Sacramento, California, United States, 95817 Contact: Site Public Contact 916-734-3089 Principal Investigator: Jonathan W. Riess United States, Colorado University of Colorado Hospital Active, not recruiting Aurora, Colorado, United States, 80045 United States, Florida Moffitt Cancer Center Recruiting Tampa, Florida, United States, 33612 Contact: Site Public Contact 800-456-7121 canceranswers@moffitt.org Principal Investigator: Eric B. Haura United States, Georgia Emory University Hospital/Winship Cancer Institute Recruiting Atlanta, Georgia, United States, 30322 Contact: Site Public Contact 404-778-1868 Principal Investigator: Suchita Pakkala United States, Kentucky University of Kentucky/Markey Cancer Center Active, not recruiting Lexington, Kentucky, United States, 40536 United States, Maryland Johns Hopkins University/Sidney Kimmel Cancer Center Recruiting Baltimore, Maryland, United States, 21287 Contact: Site Public Contact 410-955-8804 jhcccro@jhmi.edu Principal Investigator: Christine L. Hann United States, Massachusetts Massachusetts General Hospital Cancer Center Recruiting Boston, Massachusetts, United States, 02114 Contact: Site Public Contact 877-726-5130 Principal Investigator: Geoffrey R. Oxnard Dana-Farber Cancer Institute Recruiting Boston, Massachusetts, United States, 02215 Contact: Site Public Contact 877-442-3324 Principal Investigator: Geoffrey R. Oxnard United States, New Jersey Rutgers Cancer Institute of New Jersey-Robert Wood Johnson University Hospital Recruiting New Brunswick, New Jersey, United States, 08903 Contact: Site Public Contact 732-235-8675 Principal Investigator: Jyoti Malhotra Rutgers Cancer Institute of New Jersey Recruiting New Brunswick, New Jersey, United States, 08903 Contact: Site Public Contact 732-235-8675 Principal Investigator: Jyoti Malhotra United States, North Carolina UNC Lineberger Comprehensive Cancer Center Recruiting Chapel Hill, North Carolina, United States, 27599 Contact: Site Public Contact 877-668-0683 cancerclinicaltrials@med.unc.edu Principal Investigator: Jared M. Weiss Duke University Medical Center Recruiting Durham, North Carolina, United States, 27710 Contact: Site Public Contact 888-275-3853 Principal Investigator: James L. Abbruzzese United States, Ohio Ohio State University Comprehensive Cancer Center Recruiting Columbus, Ohio, United States, 43210 Contact: Site Public Contact 800-293-5066 Jamesline@osumc.edu Principal Investigator: Erin M. Bertino United States, Pennsylvania University of Pittsburgh Cancer Institute (UPCI) Recruiting Pittsburgh, Pennsylvania, United States, 15232 Contact: Site Public Contact 412-647-8073 Principal Investigator: Liza C. Villaruz United States, Virginia University of Virginia Cancer Center Recruiting Charlottesville, Virginia, United States, 22908 Contact: Site Public Contact 434-243-6303 PAS9E@virginia.edu Principal Investigator: Ryan D. Gentzler Collapse << |
| NCT02079740 | Advanced Malignant Solid Neopl... more >>asm KRAS Gene Mutation Metastatic Malignant Solid Neoplasm NRAS Gene Mutation Recurrent Colorectal Carcinoma Recurrent Lung Carcinoma Recurrent Malignant Solid Neoplasm Recurrent Pancreatic Carcinoma Stage III Colorectal Cancer AJCC v7 Stage III Lung Cancer AJCC v7 Stage III Pancreatic Cancer AJCC v6 and v7 Stage IIIA Colorectal Cancer AJCC v7 Stage IIIB Colorectal Cancer AJCC v7 Stage IIIC Colorectal Cancer AJCC v7 Stage IV Colorectal Cancer AJCC v7 Stage IV Lung Cancer AJCC v7 Stage IV Pancreatic Cancer AJCC v6 and v7 Stage IVA Colorectal Cancer AJCC v7 Stage IVB Colorectal Cancer AJCC v7 Unresectable Malignant Neoplasm Collapse << | Phase 1 Phase 2 | Recruiting | - | United States, Massachusetts ... more >> Massachusetts General Hospital Cancer Center Suspended Boston, Massachusetts, United States, 02114 Dana-Farber Cancer Institute Recruiting Boston, Massachusetts, United States, 02215 Contact: Ryan B. Corcoran 877-726-5130 Principal Investigator: Ryan B. Corcoran Collapse << |
| NCT03366103 | Metastatic Malignant Solid Neo... more >>plasm Recurrent Malignant Solid Neoplasm Recurrent Small Cell Lung Carcinoma Stage III Small Cell Lung Carcinoma AJCC v7 Stage IIIA Small Cell Lung Carcinoma AJCC v7 Stage IIIB Small Cell Lung Carcinoma AJCC v7 Stage IV Small Cell Lung Carcinoma AJCC v7 Unresectable Solid Neoplasm Collapse << | Phase 1 Phase 2 | Recruiting | May 31, 2020 | United States, Maryland ... more >> Johns Hopkins University/Sidney Kimmel Cancer Center Recruiting Baltimore, Maryland, United States, 21287 Contact: Site Public Contact 410-955-8804 jhcccro@jhmi.edu Principal Investigator: Christine L. Hann United States, Massachusetts Massachusetts General Hospital Cancer Center Recruiting Boston, Massachusetts, United States, 02114 Contact: Site Public Contact 877-726-5130 Principal Investigator: Anna F. Farago Brigham and Women's Hospital Recruiting Boston, Massachusetts, United States, 02115 Contact: Site Public Contact 888-823-5923 ctsucontact@westat.com Principal Investigator: Anna F. Farago Beth Israel Deaconess Medical Center Recruiting Boston, Massachusetts, United States, 02215 Contact: Site Public Contact 617-667-9925 Principal Investigator: Anna F. Farago Dana-Farber Cancer Institute Recruiting Boston, Massachusetts, United States, 02215 Contact: Site Public Contact 877-442-3324 Principal Investigator: Anna F. Farago United States, New Jersey Memorial Sloan Kettering Basking Ridge Recruiting Basking Ridge, New Jersey, United States, 07920 Contact: Site Public Contact 212-639-7592 Principal Investigator: Wei-Chu V. Lai Memorial Sloan Kettering Monmouth Recruiting Middletown, New Jersey, United States, 07748 Contact: Site Public Contact 212-639-7592 Principal Investigator: Wei-Chu V. Lai United States, New York Memorial Sloan Kettering Commack Recruiting Commack, New York, United States, 11725 Contact: Site Public Contact 212-639-7592 Principal Investigator: Wei-Chu V. Lai Memorial Sloan Kettering Westchester Recruiting Harrison, New York, United States, 10604 Contact: Site Public Contact 212-639-7592 Principal Investigator: Wei-Chu V. Lai Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Site Public Contact 212-639-7592 Principal Investigator: Wei-Chu V. Lai Memorial Sloan Kettering Rockville Centre Recruiting Rockville Centre, New York, United States, 11570 Contact: Site Public Contact 212-639-7592 Principal Investigator: Wei-Chu V. Lai Collapse << |
| NCT00445198 | Small Cell Lung Cancer ... more >> Small Cell Lung Carcinoma Collapse << | Phase 1 Phase 2 | Completed | - | United States, Arizona ... more >> Site Reference ID/Investigator# 13605 Peoria, Arizona, United States, 85381 Site Reference ID/Investigator# 5261 Phoenix, Arizona, United States, 85013 United States, California Site Reference ID/Investigator# 11942 Los Angeles, California, United States, 90095-7187 Site Reference ID/Investigator# 4718 Sacramento, California, United States, 95817 United States, Colorado Site Reference ID/Investigator# 3755 Aurora, Colorado, United States, 80045-0510 United States, Georgia Site Reference ID/Investigator# 8324 Atlanta, Georgia, United States, 30322 United States, Illinois Site Reference ID/Investigator# 2623 Chicago, Illinois, United States, 60612 United States, Maryland Site Reference ID/Investigator# 2625 Baltimore, Maryland, United States, 21231-1000 Site Reference ID/Investigator# 12343 Bethesda, Maryland, United States, 20892 United States, Massachusetts Site Reference ID/Investigator# 11941 Boston, Massachusetts, United States, 02215 Site Reference ID/Investigator# 2626 Boston, Massachusetts, United States, 02215 United States, North Carolina Site Reference ID/Investigator# 4934 Charlotte, North Carolina, United States, 28204 United States, Tennessee Site Reference ID/Investigator# 2624 Nashville, Tennessee, United States, 37203 United States, Washington Site Reference ID/Investigator# 6650 Tacoma, Washington, United States, 98405 Canada Site Reference ID/Investigator# 7493 Edmonton, Canada, T6G 1Z2 Site Reference ID/Investigator# 7635 Ottawa, Canada, K1H 8L6 United Kingdom Site Reference ID/Investigator# 18541 Leicester, United Kingdom, LE1 5WW Site Reference ID/Investigator# 2622 Manchester, United Kingdom, M20 4BX Collapse << |
| NCT00982566 | Lymphoid Malignancy ... more >> Solid Tumors Collapse << | Phase 1 | Completed | - | - |
| NCT00481091 | Chronic Lymphocytic Leukemia | Phase 1 Phase 2 | Active, not recruiting | September 4, 2020 | United States, California ... more >> Ucsd /Id# 5566 La Jolla, California, United States, 92037 United States, Massachusetts Dana-Farber Cancer Institute /ID# 5547 Boston, Massachusetts, United States, 02215 United States, Nebraska Univ Nebraska Med Ctr /ID# 12261 Omaha, Nebraska, United States, 68198 United States, New York North Shore University Hospital /ID# 12267 New Hyde Park, New York, United States, 11040 United States, Texas Univ TX, MD Anderson /ID# 5575 Houston, Texas, United States, 77030 United States, Washington Northwest Medical Specialties /ID# 26428 Tacoma, Washington, United States, 98405 Australia, Victoria Peter MacCallum Cancer Ctr /ID# 6583 Melbourne, Victoria, Australia, 3000 Royal Melbourne Hospital /ID# 5576 Parkville, Victoria, Australia, 3050 Germany Uniklinik Koln /ID# 5924 Köln, Nordrhein-Westfalen, Germany, 50937 United Kingdom Leicester Royal Infirmary /ID# 15081 Leicester, England, United Kingdom, LE1 5WW Collapse << |
| NCT02143401 | Cirrhosis Hep... more >>atitis B Infection Hepatitis C Infection Metastatic Malignant Solid Neoplasm Recurrent Hepatocellular Carcinoma Recurrent Malignant Solid Neoplasm Refractory Malignant Neoplasm Stage IV Hepatocellular Carcinoma AJCC v7 Unresectable Solid Neoplasm Collapse << | Phase 1 | Recruiting | - | United States, Arizona ... more >> Mayo Clinic in Arizona Recruiting Scottsdale, Arizona, United States, 85259 Contact: Site Public Contact 855-776-0015 Principal Investigator: Brian A. Costello United States, California University of California Davis Comprehensive Cancer Center Recruiting Sacramento, California, United States, 95817 Contact: Site Public Contact 916-734-3089 Principal Investigator: Edward J. Kim United States, Colorado University of Colorado Hospital Recruiting Aurora, Colorado, United States, 80045 Contact: Site Public Contact 720-848-0650 Principal Investigator: Sarah L. Davis United States, Florida Mayo Clinic in Florida Recruiting Jacksonville, Florida, United States, 32224-9980 Contact: Site Public Contact 855-776-0015 Principal Investigator: Brian A. Costello United States, Iowa University of Iowa/Holden Comprehensive Cancer Center Recruiting Iowa City, Iowa, United States, 52242 Contact: Site Public Contact 800-237-1225 Principal Investigator: Mohammed M. Milhem United States, Maryland University of Maryland/Greenebaum Cancer Center Recruiting Baltimore, Maryland, United States, 21201 Contact: Site Public Contact 800-888-8823 Principal Investigator: Edward A. Sausville United States, Minnesota Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Site Public Contact 855-776-0015 Principal Investigator: Brian A. Costello United States, New York Columbia University/Herbert Irving Cancer Center Recruiting New York, New York, United States, 10032 Contact: Site Public Contact 212-305-6361 nr2616@cumc.columbia.edu Principal Investigator: Gulam A. Manji United States, Pennsylvania Thomas Jefferson University Hospital Recruiting Philadelphia, Pennsylvania, United States, 19107 Contact: Site Public Contact 215-955-6084 Principal Investigator: James A. Posey United States, Texas M D Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Site Public Contact 877-312-3961 Principal Investigator: Ahmed O. Kaseb Collapse << |
| NCT03592576 | - | - | - | - | - |
| NCT01087151 | Chronic Lymphocytic Leukemia | Phase 2 | Completed | - | - |
| NCT03181126 | Acute Lymphoblastic Leukemia (... more >>ALL) Lymphoblastic Lymphoma Collapse << | Phase 1 | Recruiting | June 17, 2020 | United States, California ... more >> City of Hope /ID# 169029 Recruiting Duarte, California, United States, 91010 LPCH Stanford /ID# 163337 Recruiting Palo Alto, California, United States, 94304 United States, Illinois University of Chicago /ID# 163369 Recruiting Chicago, Illinois, United States, 60637 United States, Missouri Washington University-School of Medicine /ID# 165689 Recruiting Saint Louis, Missouri, United States, 63110 United States, North Carolina Univ NC Chapel Hill /ID# 163509 Recruiting Chapel Hill, North Carolina, United States, 27514-4220 United States, Ohio Cincinnati Children's Hospital /ID# 164619 Recruiting Cincinnati, Ohio, United States, 45229 Nationwide Childrens Hospital /ID# 163372 Recruiting Columbus, Ohio, United States, 43205 United States, Oregon Oregon Health and Science University /ID# 165690 Recruiting Portland, Oregon, United States, 97239-3098 United States, Tennessee St. Jude Childrens Res Hosp /ID# 163335 Recruiting Memphis, Tennessee, United States, 38105 United States, Texas UT Southwestern Medical Center /ID# 163346 Recruiting Dallas, Texas, United States, 75390-7208 MD Anderson Cancer Center /ID# 163327 Recruiting Houston, Texas, United States, 77030 United States, Wisconsin University of Wisconsin-Madiso /ID# 165691 Recruiting Madison, Wisconsin, United States, 53705 Australia, Victoria Alfred Hospital /ID# 169576 Not yet recruiting Melbourne, Victoria, Australia, 3004 Victorian Comprehensive Cancer /ID# 165710 Recruiting Melbourne, Victoria, Australia, 3050 Royal Children's Hospital /ID# 163322 Recruiting Melbourne, Victoria, Australia, 3052 Collapse << |
| NCT00406809 | Chronic Lymphoid Leukemia ... more >> Lymphoid Malignancies Non-Hodgkin's Lymphoma Follicular Lymphoma Mantle Cell Lymphoma Peripheral T-cell Lymphoma Collapse << | Phase 1 Phase 2 | Completed | - | United States, California ... more >> Site Reference ID/Investigator# 4997 Los Angeles, California, United States, 90033 Site Reference ID/Investigator# 9104 Los Angeles, California, United States, 90095 United States, Maryland Site Reference ID/Investigator# 2613 Bethesda, Maryland, United States, 20892 United States, Massachusetts Site Reference ID/Investigator# 40243 Boston, Massachusetts, United States, 02215 Site Reference ID/Investigator# 4745 Boston, Massachusetts, United States, 02215 United States, New York Site Reference ID/Investigator# 2628 Buffalo, New York, United States, 14263 Site Reference ID/Investigator# 23543 New York, New York, United States, 10016 Site Reference ID/Investigator# 2627 New York, New York, United States, 10021 Site Reference ID/Investigator# 2614 New York, New York, United States, 10032 Site Reference ID/Investigator# 5383 New York, New York, United States, 10065 Site Reference ID/Investigator# 12306 Rochester, New York, United States, 14642 Canada Site Reference ID/Investigator# 8941 Edmonton, Canada, T6G 1Z2 Collapse << |
| NCT01828476 | Prostate Cancer | Phase 2 | Terminated(The Investigator le... more >>ft the organization.) Collapse << | - | United States, New Jersey ... more >> Rutgers Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08901 Collapse << |
| NCT00743028 | Chronic Lymphocytic Leukemia ... more >> Lymphomas Leukemias Collapse << | Phase 1 | Completed | - | United States, California ... more >> Site Reference ID/Investigator# 10281 Encinitas, California, United States, 92024 Site Reference ID/Investigator# 10282 Santa Monica, California, United States, 90404 United States, Maryland Site Reference ID/Investigator# 16341 Bethesda, Maryland, United States, 20892 United States, New Hampshire Site Reference ID/Investigator# 9441 Lebanon, New Hampshire, United States, 03756 United States, New Jersey Site Reference ID/Investigator# 20041 Hackensack, New Jersey, United States, 07601 Collapse << |
| NCT01828476 | - | - | Terminated(The Investigator le... more >>ft the organization.) Collapse << | - | - |
| NCT00888108 | Solid Tumor | Phase 1 | Completed | - | United States, Arizona ... more >> Site Reference ID/Investigator# 51982 Scottsdale, Arizona, United States, 85258 United States, Florida Site Reference ID/Investigator# 44182 Fort Lauderdale, Florida, United States, 33308 United States, Maryland Site Reference ID/Investigator# 43962 Baltimore, Maryland, United States, 21231 Netherlands Site Reference ID/Investigator# 12844 Rotterdam, Netherlands, 3015 CE Site Reference ID/Investigator# 20042 Rotterdam, Netherlands, 3015 CE United Kingdom Site Reference ID/Investigator# 12845 Surrey, United Kingdom, SM2 5PT Collapse << |
| NCT01557777 | Chronic Lymphocytic Leukemia | Phase 2 | Completed | - | United States, California ... more >> Site Reference ID/Investigator# 74036 La Jolla, California, United States, 92093-0698 Site Reference ID/Investigator# 74035 Los Angeles, California, United States, 90095-7059 Site Reference ID/Investigator# 74038 Pleasant Hill, California, United States, 94523 Site Reference ID/Investigator# 74039 Santa Maria, California, United States, 93454 United States, Maryland Site Reference ID/Investigator# 74033 Bethesda, Maryland, United States, 20817 Australia Site Reference ID/Investigator# 79201 Coburg, Australia, 3058 Site Reference ID/Investigator# 78993 Greenslopes, Australia, 4120 Israel Site Reference ID/Investigator# 77860 Afula, Israel, 18101 Site Reference ID/Investigator# 77637 Rechovot, Israel, 76100 Poland Site Reference ID/Investigator# 77413 Gdansk, Poland, 80-952 Ukraine Site Reference ID/Investigator# 79203 Ivano-Frankivsk, Ukraine, 76008 Site Reference ID/Investigator# 79205 Khmelnitsky, Ukraine, 29000 Site Reference ID/Investigator# 79202 Kyiv, Ukraine, 03115 Site Reference ID/Investigator# 79204 Lviv, Ukraine, 79044 Site Reference ID/Investigator# 79206 Poltava, Ukraine, 36024 Collapse << |
| NCT00868413 | Chronic Lymphocytic Leukemia | Phase 1 | Completed | - | United States, California ... more >> Site Reference ID/Investigator# 17841 La Jolla, California, United States, 92093 Site Reference ID/Investigator# 25899 Stanford, California, United States, 94305-5821 United States, Maryland Site Reference ID/Investigator# 21622 Baltimore, Maryland, United States, 21231-1000 United States, Ohio Site Reference ID/Investigator# 21621 Columbus, Ohio, United States, 43210 United States, Pennsylvania Site Reference ID/Investigator# 39613 Philadelphia, Pennsylvania, United States, 19111 United States, Texas Site Reference ID/Investigator# 17943 Houston, Texas, United States, 77030-4009 Collapse << |
| NCT01121133 | Lymphoma, Including Chronic Ly... more >>mphocytic Leukemia Solid Tumors Collapse << | Phase 1 | Completed | - | - |
| NCT00918450 | B-cell Chronic Lymphocytic Leu... more >>kemia Collapse << | Phase 2 | Withdrawn(Sponsor has decided ... more >>to not proceed with this study.) Collapse << | - | - |
| NCT00788684 | CD20-Positive Lymphoid Maligna... more >>ncies Chronic Lymphoid Leukemia Hematological Malignancies Non-Hodgkin's Lymphoma Collapse << | Phase 1 | Active, not recruiting | April 30, 2020 | United States, Arizona ... more >> University Arizona Cancer Ctr /ID# 16721 Tucson, Arizona, United States, 85719-1478 United States, California Stanford Univ School of Med /ID# 9782 Stanford, California, United States, 94305-2200 United States, Ohio Cleveland Clinic Foundation /ID# 9784 Cleveland, Ohio, United States, 44195 United States, Wisconsin Univ of Wisconsin Hosp/Clinics /ID# 21701 Madison, Wisconsin, United States, 53792-0001 Australia, Victoria Peter MacCallum Cancer Ctr /ID# 25067 Melbourne, Victoria, Australia, 3000 Royal Melbourne Hospital /ID# 9781 Parkville, Victoria, Australia, 3050 Collapse << |
| NCT01009073 | Solid Tumors | Phase 1 | Completed | - | United States, California ... more >> Site Reference ID/Investigator# 37463 Santa Monica, California, United States, 90404 United States, Michigan Site Reference ID/Investigator# 36342 Detroit, Michigan, United States, 48201 United States, Texas Site Reference ID/Investigator# 24046 San Antonio, Texas, United States, 78229 United States, Washington Site Reference ID/Investigator# 44917 Tacoma, Washington, United States, 98405 Collapse << |
| NCT00891605 | Solid Tumors | Phase 1 | Completed | - | - |
| NCT00887757 | Solid Tumors | Phase 1 | Completed | - | - |
| NCT01021358 | Lymphoma Chro... more >>nic Lymphocytic Leukemia Solid Tumors Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> Site Reference ID/Investigator# 25068 San Antonio, Texas, United States, 78229 Collapse << |
| NCT03222609 | Myelofibrosis (MF) | Phase 2 | Recruiting | June 11, 2021 | United States, Alabama ... more >> UAB Comprehensive Cancer Cente /ID# 165464 Recruiting Birmingham, Alabama, United States, 35217 United States, California Ucsd /Id# 164084 Recruiting La Jolla, California, United States, 92037 Usc /Id# 164095 Recruiting Los Angeles, California, United States, 90033 United States, Florida Mayo Clinic /ID# 164201 Recruiting Jacksonville, Florida, United States, 32224 Moffitt Cancer Center /ID# 164082 Recruiting Tampa, Florida, United States, 33612 United States, Illinois University of Chicago /ID# 164115 Recruiting Chicago, Illinois, United States, 60637 United States, Indiana Indiana Blood & Marrow Transpl /ID# 165075 Recruiting Indianapolis, Indiana, United States, 46237 United States, Massachusetts Dana-Farber Cancer Institute /ID# 162675 Recruiting Boston, Massachusetts, United States, 02215 United States, Michigan Henry Ford Hospital /ID# 162682 Recruiting Detroit, Michigan, United States, 48202 United States, New York Weill Cornell Medical College /ID# 162679 Recruiting New York, New York, United States, 10021 United States, Texas MD Anderson Cancer Center /ID# 162683 Recruiting Houston, Texas, United States, 77030 UT Health Cancer Center /ID# 164094 Recruiting San Antonio, Texas, United States, 78229 United States, Utah University of Utah /ID# 164116 Recruiting Salt Lake City, Utah, United States, 84112-5500 United Kingdom Guy's and St Thomas' NHS Found /ID# 164110 Recruiting London, London, City Of, United Kingdom, SE1 9RT Christie NHS Foundation Trust /ID# 164111 Recruiting Manchester, United Kingdom, M20 4BX Collapse << |
| NCT00878449 | Solid Tumors | Phase 1 | Completed | - | United States, Illinois ... more >> Site Reference ID/Investigator# 13323 Chicago, Illinois, United States, 60637 Site Reference ID/Investigator# 12841 Maywood, Illinois, United States, 60153 United States, Maryland Site Reference ID/Investigator# 12303 Baltimore, Maryland, United States, 21231 United States, Massachusetts Site Reference ID/Investigator# 12305 Boston, Massachusetts, United States, 02215 Site Reference ID/Investigator# 20381 Boston, Massachusetts, United States, 02215 United States, Michigan Site Reference ID/Investigator# 43505 Detroit, Michigan, United States, 48202 United States, New Jersey Site Reference ID/Investigator# 13322 New Brunswick, New Jersey, United States, 08901 Collapse << |
| NCT02591095 | Platinum-resistant or Refracto... more >>ry Ovarian Cancer Collapse << | Phase 2 | Active, not recruiting | December 2018 | France ... more >> CHU Besançon - Hôpital Jean Minjoz Besançon, France Institut Bergonié Bordeaux, France Centre Francois Baclesse Caen, France Centre Oscar Lambret Lille, France Centre Léon Bérard Lyon, France CHU Lyon, France ICM Val d'Aurelle Montpellier, France ICL Institut de Cancérologie de Lorraine Nancy, France Centre Catherine de Sienne Nantes, France ICO Centre René Gauducheau Nantes, France ICO Paul Papin Nantes, France Centre Antoine LACASSAGNE Nice, France Hôpital Européen Georges Pompidou Paris, France Hôpital Tenon Paris, France Institut Claudius Regaud Toulouse, France Gustave Roussy Villejuif, France Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| Bcl-2 | Ki:1nM | |
| Bcl-w | Ki:1nM | |
| Bcl-xL | Ki:0.5nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
