| NCT03551522 | NASH - Nonalcoholic Steatohepa... more >>titis Collapse << | Phase 2 | Recruiting | December 31, 2020 | United States, Arizona
... more >>
CymaBay Research Site
Recruiting
Glendale, Arizona, United States, 85306
CymaBay Research Site
Recruiting
Tucson, Arizona, United States, 85712
United States, California
CymaBay Research Site
Recruiting
Los Angeles, California, United States, 90057
United States, Florida
CymaBay Research Site
Recruiting
Boca Raton, Florida, United States, 33434
CymaBay Research Site
Recruiting
Lakewood Ranch, Florida, United States, 34211
United States, Mississippi
CymaBay Research Site
Recruiting
Flowood, Mississippi, United States, 39232
United States, Tennessee
CymaBay Research Site
Recruiting
Clarksville, Tennessee, United States, 37040
CymaBay Research Site
Recruiting
Germantown, Tennessee, United States, 38138
CymaBay Research Site
Recruiting
Hermitage, Tennessee, United States, 37076
CymaBay Research Site
Recruiting
Knoxville, Tennessee, United States, 37909
United States, Texas
CymaBay Research Site
Recruiting
Chandler, Texas, United States, 85224
CymaBay Research Site
Recruiting
Dallas, Texas, United States, 75246
CymaBay Research Site
Recruiting
Live Oak, Texas, United States, 78233
CymaBay Research Site
Recruiting
Rollingwood, Texas, United States, 78746 Collapse << |
| NCT03602560 | Primary Biliary Cholangitis | Phase 3 | Recruiting | December 2021 | United States, Arizona
... more >>
Institute for Liver Health
Recruiting
Chandler, Arizona, United States, 85224
Contact: Dana Robledo 480-686-8874 drobledo@iliverhealth.org
Principal Investigator: Anita Kohli, MD
United States, Colorado
Univeristy of Colorado Denver and Hospital
Recruiting
Aurora, Colorado, United States, 80045
Contact: Joselin Gonzalez-Diaz 303-724-1866 joselin.gonzalez-diaz@ucdenver.edu
Principal Investigator: Lisa Forman, MD
United States, Florida
Excel Medical Clinical Trials, LLC
Recruiting
Boca Raton, Florida, United States, 33434
Contact: Jennifer Olivar 561-756-8206 jolivar@emclinicaltrials.com
Principal Investigator: Seth J Baum, MD
Florida Reserach Institute
Recruiting
Lakewood Ranch, Florida, United States, 34211
Contact: Kalina Miller 941-727-7772 kalina.miller@fdhs.com
Principal Investigator: Guy W Neff, MD
United States, Georgia
Digestive Healthcare of Georgia
Recruiting
Atlanta, Georgia, United States, 30309
Contact: Debeh Legaspi 678-686-5073 dlegaspi@digestivehealthcare.net
Principal Investigator: Michael R Galambos, MD
United States, Indiana
Indianapolis Gastroenterology Research Foundation
Recruiting
Indianapolis, Indiana, United States, 46237
Contact: Sheila Thompson 317-865-2959 sthompson@indygastro.com
Principal Investigator: David C Pound, MD
United States, Minnesota
Minnesota Gastroenterlogy, P.A.
Recruiting
Maplewood, Minnesota, United States, 55117
Contact: Gavin Mark 612-870-5597 gavin.mark@mngastro.com
Principal Investigator: Donald L Zogg, MD
United States, Mississippi
Southern Therapy and Advance Research (STAR) LLC
Recruiting
Jackson, Mississippi, United States, 39216
Contact: Evelyn Arendale 769-251-5674 earendale@jacksonliverandgi.com
Principal Investigator: Brian B Borg, MD
United States, Missouri
Kansas City Research Institute
Recruiting
Kansas City, Missouri, United States, 64131
Contact: Kelly Vargas 816-759-5274 kvargas@kcresearchinstitute.com
Contact: Jeanne Zirkle (816) 759-5274 jzirkle@kcresearchinstitute.com
Principal Investigator: Bradley L Freilich, MD
United States, New York
Concorde Medical Group
Recruiting
New York, New York, United States, 10016
Contact: Resmi Varughese 212-726-9472 rvarughese@concordemed.com
Principal Investigator: Hillel Tobias, MD
NYU Langone Health
Recruiting
New York, New York, United States, 10016
Contact: Kristyn Pierce 646-501-8468 Kristyn.Pierce@nyulangone.org
Principal Investigator: Carmen Stanca, MD
University of Rochester Medical Center
Recruiting
Rochester, New York, United States, 14642
Contact: Juliana Posato 585-275-4711 juliana_posato@urmc.rochester.edu
Principal Investigator: Jonathan C Huang, DO
United States, North Carolina
Asheville Gastroenterology Associates, a Division of Digestive Health Partners, P.A.
Recruiting
Asheville, North Carolina, United States, 28801
Contact: Amber Bartlett 828-254-0881 abartlett@ncdhp.com
Principal Investigator: Adam S Zivony, MD
United States, Pennsylvania
Northeast Clinical Research Center, LLC
Recruiting
Bethlehem, Pennsylvania, United States, 18017
Contact: Jennifer Jones 610-433-4100 jennifer.jones@necresearch.org
Principal Investigator: Adam Peyton, DO
United States, Tennessee
Gastro One
Recruiting
Germantown, Tennessee, United States, 38138
Contact: Keosha Henderson 901-682-6540 khenderson@gastro1.com
Principal Investigator: Ziad Younes, MD
GIA Clinical Trials, LLC
Recruiting
Knoxville, Tennessee, United States, 37909
Contact: Kathy Karnes 865-558-0687 kkarnes@gihealthcare.com
Principal Investigator: Jason Huffman, MD
United States, Texas
Texas Digestive Disease Consultants
Recruiting
Fort Worth, Texas, United States, 76104
Contact: Neal Bhakta 817-310-4478 nbhakta@tddctx.com
Principal Investigator: Apurva A Modi, MD
Baylor College of Medicine
Recruiting
Houston, Texas, United States, 77030
Contact: Jignesh Patel 713-798-5242 jignesh.patel@bcm.edu
Principal Investigator: John M Vierling, MD
American Research Corporation at Texas Liver Institute
Recruiting
San Antonio, Texas, United States, 78215
Contact: MaryLou Hernandez 210-253-3426 mhernandez@txliver.com
Principal Investigator: Eric J Lawitz, MD
Pinnacle Clinical Research
Recruiting
San Antonio, Texas, United States, 78229
Contact: Kathy Marotta 210-982-0320 ext 1437 kmarotta@pinnacleresearch.com
Principal Investigator: Stephen Harrison, MD
United States, Virginia
Bon Secours Richmond Community Hospital
Recruiting
Richmond, Virginia, United States, 23226
Contact: Susan Vollum 804-977-8921 susan_vollum@bshsi.org
Principal Investigator: Mitchell L Shiffman, MD Collapse << |
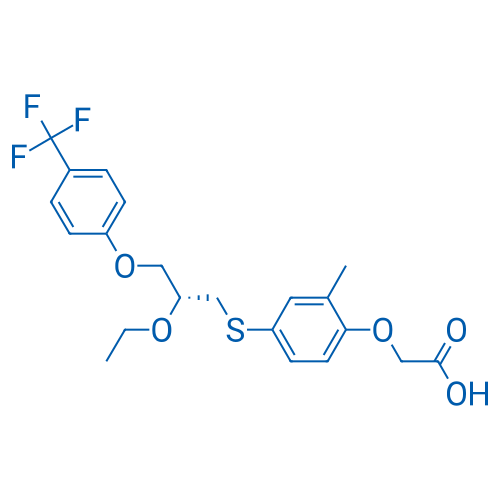
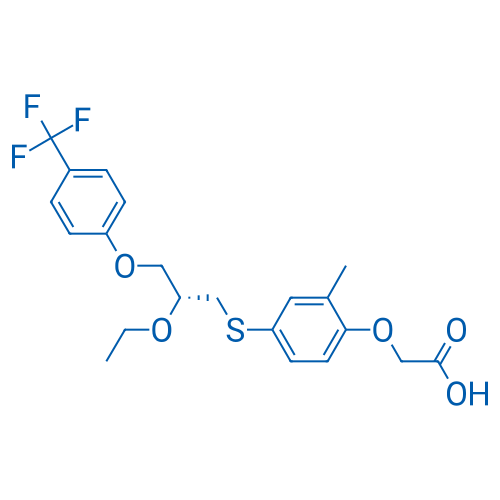

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
