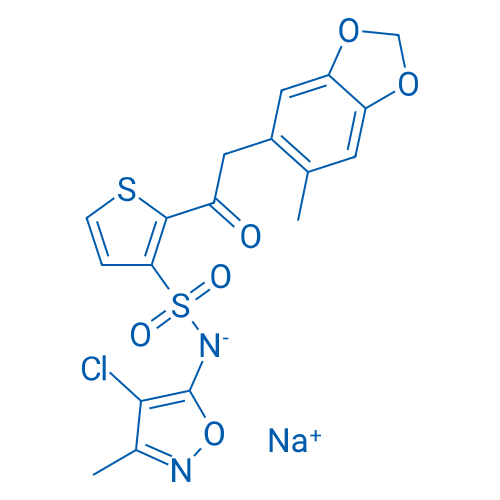
CAS No.: 210421-74-2
Sitaxsentan sodium Catalog No. CSN12974
Synonyms: IPI 1040 sodium;TBC11251 sodium;TBC11251 Sodium Salt
Sitaxsentan sodium has selectory antagonism of endothelin A (ETA) receptor with the IC50 and Ki value of 1.4 nM and 0.43 nM respectively.
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 Endothelin Receptor A
IC50:1.4nM- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00593905 - - Unknown July 2013 United States, Pennsylvania ... more >> Allegheny General Hospital Recruiting Pittsburgh, Pennsylvania, United States, 15212 Contact: Andrea L Nowicki, BA 412-359-3653 anowicki@wpahs.org Contact: Raymond L Benza, MD 412.359.3584 rbenza@wpahs.org Principal Investigator: Raymond L Benza, MD Collapse << NCT00810732 Chronic Kidney Disease ... more >> CRD Collapse << Phase 2 Completed - United Kingdom ... more >> Pfizer Investigational Site Edinburgh, Scotland, United Kingdom, EH4 2XU Collapse << NCT00796666 Pulmonary Arterial Hypertensio... more >>n Pulmonary Hypertension Collapse << Phase 3 Terminated(Safety Issue The tr... more >>ial was prematurely terminated on Dec 9, 2010, due to safety concerns, specifically new emerging evidence of hepatic injury.) Collapse << - - - 更多
- 参考文献
- [1] Tilton RG, Munsch CL, et al. Attenuation of pulmonary vascular hypertension and cardiac hypertrophy with sitaxsentan sodium, an orally active ET(A) receptor antagonist. Pulm Pharmacol Ther. 2000;13(2):87-97.
- [2] Wu C, Chan MF, et al. Discovery of TBC11251, a potent, long acting, orally active endothelin receptor-A selective antagonist. J Med Chem. 1997 May 23;40(11):1690-7.
- [3] Sharifat N, Mohammad Zadeh G, Ghaffari MA, Dayati P, Kamato D, Little PJ, Babaahmadi-Rezaei H. Endothelin-1 (ET-1) stimulates carboxy terminal Smad2 phosphorylation in vascular endothelial cells by a mechanism dependent on ET receptors and de novo protein synthesis. J Pharm Pharmacol. 2017 Jan;69(1):66-72. doi: 10.1111/jphp.12654. Epub 2016 Dec 1. PMID: 27905105.
- [4] Wu C, Decker ER, Blok N, Li J, Bourgoyne AR, Bui H, Keller KM, Knowles V, Li W, Stavros FD, Holland GW, Brock TA, Dixon RA. Acyl substitution at the ortho position of anilides enhances oral bioavailability of thiophene sulfonamides: TBC3214, an ETA selective endothelin antagonist. J Med Chem. 2001 Apr 12;44(8):1211-6. doi: 10.1021/jm000349x. PMID: 11312921.
- [5] Waxman AB. A review of sitaxsentan sodium in patients with pulmonary arterial hypertension. Vasc Health Risk Manag. 2007;3(1):151-7. PMID: 17583185; PMCID: PMC1994033.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 210421-74-2 | 储存条件 |
|
|||
| 分子式 | C18H14ClN2NaO6S2 | 运输 | 蓝冰 | |||
| 分子量 | 476.89 | 别名 | IPI 1040 sodium;TBC11251 sodium;TBC11251 Sodium Salt;Sitaxentan (sodium salt);TBC-11251;Sitaxentan sodium | |||
| 溶解度 |
|
动物实验配方 |
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
