| NCT00762424 | - | - | Completed | - | - |
| NCT00884104 | Overactive Bladder | Phase 4 | Completed | - | Korea, Republic of
... more >>
Cheon-ju, Korea, Republic of
Choongju, Korea, Republic of
Daegu, Korea, Republic of
Daejeon, Korea, Republic of
Kangneung, Korea, Republic of
Kwang-ju, Korea, Republic of
Pusan, Korea, Republic of
Seoul, Korea, Republic of
Suwon, Korea, Republic of Collapse << |
| NCT01021332 | Lower Urinary Tract Symptoms
... more >> Benign Prostatic Hyperplasia Collapse << | Phase 3 | Completed | - | - |
| NCT00762424 | Kidney Stone | Not Applicable | Completed | - | United States, Pennsylvania
... more >>
York Hospital Emergency Department
York, Pennsylvania, United States, 17405 Collapse << |
| NCT02033798 | Chronic Kidney Failure
... more >> Prostatic Hyperplasia Collapse << | Not Applicable | Unknown | June 2016 | Korea, Republic of
... more >>
St. Vincent's Hospital
Not yet recruiting
Suwon, Gyeonggi-do, Korea, Republic of, 442-723
Contact: Seung-Ju Lee Lee, MD, PhD 82-31-249-8305 lee.seungju@gmail.com Collapse << |
| NCT01018511 | - | - | Completed | - | - |
| NCT01021332 | - | - | Completed | - | - |
| NCT00831701 | Ureteral Calculi | Not Applicable | Completed | - | Switzerland
... more >> Department of Urology, University Hospital Zürich, Switzerland
Zurich, Switzerland, 8091 Collapse << |
| NCT01018511 | Benign Prostatic Hyperplasia
... more >> Lower Urinary Tract Symptoms Collapse << | Phase 3 | Completed | - | - |
| NCT02058368 | Prostatic Hyperplasia | Phase 3 | Completed | - | - |
| NCT00831701 | - | - | Completed | - | - |
| NCT03027115 | Hernia
Urinar... more >>y Retention Collapse << | Phase 4 | Recruiting | January 27, 2020 | United States, Georgia
... more >>
Memorial Health University Medical Center
Recruiting
Savannah, Georgia, United States, 31404
Contact: Vishu Danthuluri, M.D. 256-665-5214 vishwanathdanthuluri@memorialhealth.com
Contact: Andrew Schwemmer, M.D. SchweAn2@memorialhealth.com Collapse << |
| NCT01294592 | Prostatic Hyperplasia | Phase 4 | Completed | - | - |
| NCT01560091 | Kidney Stones | Phase 3 | Withdrawn(accrual of subjects ... more >>did not occur as anticipated) Collapse << | - | United States, Pennsylvania
... more >>
Albert Einstein Healthcare Network
Philadelphia, Pennsylvania, United States, 19141 Collapse << |
| NCT01010048 | Urinary Calculus | Phase 4 | Unknown | December 2010 | China, Chongqing
... more >>
The First Affiliated Hospital, ChongQing medical University
Recruiting
ChongQing, Chongqing, China, 400016
Contact: Yunfeng He, Doctor 86-23-89011121
Contact: Xiaohou Wu, Doctor 86-23-89011122 Collapse << |
| NCT00606983 | Rectal Cancer
... more >> Urinary Retention Collapse << | Phase 3 | Completed | - | Korea, Republic of
... more >>
Department of Surgery, Seoul National University Bundang Hospital
Seongnam, Korea, Republic of, 463-707 Collapse << |
| NCT00209131 | Urolithiasis | Not Applicable | Terminated(Due to frequent tur... more >>nover of research coordinators and thus poor study accrual.) Collapse << | - | United States, Georgia
... more >>
Emory University
Atlanta, Georgia, United States, 30322 Collapse << |
| NCT02247505 | Healty Male Volunteers | Phase 1 | Completed | - | Korea, Republic of
... more >>
Chonbuk National University Hospital
Jeonju-si, Korea, Republic of Collapse << |
| NCT00209131 | - | - | Terminated(Due to frequent tur... more >>nover of research coordinators and thus poor study accrual.) Collapse << | - | - |
| NCT02417831 | Prostatic Hyperplasia | Phase 1 | Completed | - | South Africa
... more >> Boehringer Ingelheim Investigational Site
Bloemfontein, South Africa Collapse << |
| NCT01294592 | - | - | Completed | - | - |
| NCT00379067 | Benign Prostatic Hyperplasia | Phase 4 | Completed | - | - |
| NCT01167062 | Ureteral Calculi | Phase 4 | Unknown | - | Thailand
... more >> Maharat Nakhon Chiangmai Hospital
Recruiting
Muang, Chiangmai, Thailand, 50200
Contact: Bannakij Lojanapiwat, M.D. 66-53-945532 blojanap@mail.med.cmu.ac.th
Principal Investigator: Bannakij Lojanapiwat, M.D. Collapse << |
| NCT00478998 | Upper Tract Ureterolithiasis | Phase 2 | Unknown | - | Israel
... more >> Hadassah Medical Organization
Not yet recruiting
Jerusalem, Israel, 91120
Contact: Mordechai Duvdevani, MD 0508946816 moti_duv@yahoo.com
Principal Investigator: Mordechai Duvdevani, MD Collapse << |
| NCT00340704 | Bladder, Neurogenic | Phase 2 | Completed | - | - |
| NCT01726270 | Lower Urinary Tract Symptoms | Phase 2 | Completed | - | United States, Alabama
... more >>
527.82.170 Boehringer Ingelheim Investigational Site
Hoover, Alabama, United States
United States, Arizona
527.82.167 Boehringer Ingelheim Investigational Site
Mesa, Arizona, United States
United States, California
527.82.177 Boehringer Ingelheim Investigational Site
Encino, California, United States
527.82.168 Boehringer Ingelheim Investigational Site
Garden Grove, California, United States
527.82.171 Boehringer Ingelheim Investigational Site
La Habra, California, United States
United States, Minnesota
527.82.161 Boehringer Ingelheim Investigational Site
Anoka, Minnesota, United States
527.82.179 Boehringer Ingelheim Investigational Site
Elk River, Minnesota, United States
527.82.163 Boehringer Ingelheim Investigational Site
Fridley, Minnesota, United States
527.82.165 Boehringer Ingelheim Investigational Site
Roseville, Minnesota, United States
527.82.166 Boehringer Ingelheim Investigational Site
St. Francis, Minnesota, United States
United States, New Mexico
527.82.176 Boehringer Ingelheim Investigational Site
Albuquerque, New Mexico, United States
United States, Texas
527.82.181 Boehringer Ingelheim Investigational Site
Longview, Texas, United States
527.82.160 Boehringer Ingelheim Investigational Site
Tyler, Texas, United States
United States, Utah
527.82.162 Boehringer Ingelheim Investigational Site
Bountiful, Utah, United States
527.82.180 Boehringer Ingelheim Investigational Site
Layton, Utah, United States
527.82.173 Boehringer Ingelheim Investigational Site
Salt Lake City, Utah, United States
527.82.174 Boehringer Ingelheim Investigational Site
Syracuse, Utah, United States
United States, Virginia
527.82.169 Boehringer Ingelheim Investigational Site
Richmond, Virginia, United States Collapse << |
| NCT00913315 | Prostatitis | Not Applicable | Unknown | January 2010 | China, Chongqing
... more >>
Department of Urology, the First Affiliated Hospital of Chongqing Medical University
Not yet recruiting
Chongqing, Chongqing, China, 400016
Contact: Xiaohou Wu, MD 86-2389011122 93404840@qq.com Collapse << |
| NCT02634489 | Phase 1
Healt... more >>hy
Benign Prostatic Hyperplasia
Lower Urinary Tract Symptoms Collapse << | Phase 1 | Completed | - | Netherlands
... more >> Site NL1
Zuidlaren, Drente, Netherlands, 9471 GP Collapse << |
| NCT01568918 | Postoperative Urinary Retentio... more >>n Collapse << | Phase 3 | Recruiting | December 2018 | United States, Minnesota
... more >>
Mayo Clinic in Rochester
Recruiting
Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00340704 | - | - | Completed | - | - |
| NCT02245490 | Prostatic Hyperplasia | Phase 4 | Completed | - | - |
| NCT01657851 | Prostatic Hyperplasia | Phase 1 | Completed | - | Russian Federation
... more >>
GSK Investigational Site
Reutov, Moscow Region, Russian Federation, 143964 Collapse << |
| NCT01533597 | Overactive Bladder | Phase 4 | Completed | - | Korea, Republic of
... more >>
Soonchunhyang University Bucheon Hospital
Bucheon, Gyeonggi-do, Korea, Republic of, 420-767 Collapse << |
| NCT01100021 | Erectile Dysfunction | Phase 1 | Completed | - | United States, Arizona
... more >>
MDS Pharma Services Inc.,
Tempe, Arizona, United States, 85283 Collapse << |
| NCT01076309 | - | - | Completed | - | Denmark
... more >> Frederiksberg University Hospital
Frederiksberg, Denmark, 2000 Collapse << |
| NCT02184585 | Prostatic Hyperplasia | Phase 1 | Completed | - | United States, Maryland
... more >>
GSK Investigational Site
Baltimore, Maryland, United States, 21225 Collapse << |
| NCT02244229 | Prostatic Hyperplasia | Phase 4 | Completed | - | - |
| NCT01533597 | - | - | Completed | - | - |
| NCT02244294 | Prostate Hyperplasia | Phase 2 | Completed | - | - |
| NCT02245503 | - | - | Completed | - | - |
| NCT02244281 | Lower Urinary Tract Symptoms | Phase 2 | Completed | - | - |
| NCT02245555 | - | - | Completed | - | - |
| NCT00510406 | Lower Urinary Tract Symptoms
... more >> Prostatic Hyperplasia Collapse << | Phase 2 | Completed | - | - |
| NCT02244255 | Prostatic Hyperplasia | Phase 4 | Completed | - | - |
| NCT02573311 | Urological Manifestations | Phase 3 | Completed | - | - |
| NCT03384524 | Diabetic Macular Edema | Phase 1
Phase 2 | Not yet recruiting | May 2019 | - |
| NCT00090103 | Prostatic Hyperplasia | Phase 3 | Completed | - | - |
| NCT03280420 | BPH With Urinary Obstruction | Not Applicable | Completed | - | Egypt
... more >> Faculty of medicine ,Al Azhar university
Cairo, El Mokhayam El Daem St.، Ezbet El-Arab, Nasr City, Cairo, Egypt Collapse << |
| NCT02180997 | LUTS
Benign P... more >>rostatic Hyperplasia
Overactive Bladder Collapse << | Phase 1 | Completed | - | Korea, Republic of
... more >>
Seoul National University Hospital(SNUH)
Seoul, Jongno-Gu, Korea, Republic of, 110-744 Collapse << |
| NCT02180789 | Lower Urinary Tract Symptoms (... more >>LUTS) Associated With Benign Prostatic Hyperplasia (BPH) Collapse << | Phase 4 | Completed | - | Taiwan
... more >>
Linkou, Taiwan Collapse << |
| NCT01953848 | EC905
Pharmac... more >>okinetics
Multiple Dose
Healthy Subjects Collapse << | Phase 1 | Completed | - | United Kingdom
... more >> Parexel Early Phase Clinical Unit
Harrow, United Kingdom, H1 3UJ Collapse << |
| NCT02266524 | Urination Disorders | Phase 1 | Completed | - | - |
| NCT01495026 | Prostatic Hyperplasia | Phase 1 | Completed | - | Australia, New South Wales
... more >>
GSK Investigational Site
Randwick, New South Wales, Australia, 2031 Collapse << |
| NCT00090103 | - | - | Completed | - | - |
| NCT02757768 | Benign Prostatic Hyperplasia
... more >> Overactive Bladder Collapse << | Phase 4 | Completed | - | - |
| NCT03144596 | Benign Prostatic Hyperplasia
... more >> Pupil Anomaly
Choroid Disease Collapse << | Phase 4 | Completed | - | - |
| NCT01215708 | Renal Calculi | Phase 3 | Unknown | December 2010 | Brazil
... more >> Clinics Hospital of University of Sao Paulo
Sao Paulo, Brazil, 05403900 Collapse << |
| NCT00507455 | Lower Urinary Tract Symptoms
... more >> Bladder Outlet Obstruction Collapse << | Phase 2 | Completed | - | - |
| NCT00796614 | Bladder, Neurogenic | Phase 2
Phase 3 | Completed | - | - |
| NCT03229889 | Nephrolithiasis | Phase 4 | Enrolling by invitation | June 1, 2019 | United States, California
... more >>
UC Irvine Health
Orange, California, United States, 92868 Collapse << |
| NCT01209988 | Prostatic Neoplasms, Prostatec... more >>tomy Collapse << | Not Applicable | Unknown | December 2011 | Korea, Republic of
... more >>
Asan Medical Center
Recruiting
Seoul, Korea, Republic of, 138-736
Contact: Choung-Soo Kim, M.D. cskim@amc.seoul.kr Collapse << |
| NCT00507455 | - | - | Completed | - | - |
| NCT01489696 | Cardiovascular
... more >> Healthy Subjects
Pharmacokinetics Collapse << | Phase 1 | Completed | - | United Kingdom
... more >> Covance CRU Ltd
Leeds, United Kingdom, LS2 9LH Collapse << |
| NCT01747993 | Urinary Retention | Phase 3 | Terminated(low inclusion) | - | France
... more >> Service de médecine interne, Hôpital Tenon
Paris, France, 75020 Collapse << |
| NCT00796614 | - | - | Completed | - | - |
| NCT02715024 | Lower Urinary Tract Symptoms
... more >> Overactive Bladder
Benign Prostatic Hyperplasia (BPH) Collapse << | Phase 4 | Completed | - | Taiwan
... more >> Site
Taipei, Taiwan Collapse << |
| NCT02483793 | Disorder of Urinary Stent | Not Applicable | Unknown | December 2016 | - |
| NCT01880619 | Relieve of Ureteral Stent Symp... more >>toms Collapse << | Phase 4 | Completed | - | Egypt
... more >> Urology and Nephrology Center
Mansoura, Egypt, 35516 Collapse << |
| NCT01943487 | Drug-Drug Interaction (DDI)
... more >> Healthy Subjects Collapse << | Phase 1 | Completed | - | France
... more >> SGS Aster
Paris, France, 75015 Collapse << |
| NCT00600405 | Nephrolithiasis
... more >> Ureteral Calculi Collapse << | Not Applicable | Completed | - | United States, Maine
... more >>
Maine Medical Center
Portland, Maine, United States, 04102 Collapse << |
| NCT02486653 | Urinary Retention
... more >> Urinary Tract Infections Collapse << | Phase 3 | Active, not recruiting | September 2018 | United States, Wisconsin
... more >>
University of Wisconsin Hospital and Clinics
Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00151567 | Ureterolithiasis
... more >> Ureteral Calculi Collapse << | Phase 3 | Completed | - | France
... more >> Service d'Urologie- Hôpital du Val de Grâce
Paris, France, 75005
Service d'Urologie - Hôpital de La Milétrie
Poitiers, France, 86021
Hôpital de Redon
Redon, France, 35600
Service d'Urologie- Hôpital Robert Debré
Reims, France, 51092
Service d'Urologie- Hôpital Pontchaillou
Rennes, France, 35033
Service d'Urologie - Hôpital Bretonneau
Tours, France, 37044 Collapse << |
| NCT02266511 | Healthy | Phase 1 | Completed | - | - |
| NCT02494349 | LUTS(Lower Urinary Tract Sympt... more >>oms)
Benign Prostatic Hyperplasia
Overactive Bladder Collapse << | Phase 1 | Completed | - | - |
| NCT02417844 | Prostatic Hyperplasia | Phase 1 | Completed | - | South Africa
... more >> Boehringer Ingelheim Investigational Site
Bloemfontein, South Africa Collapse << |
| NCT01534351 | - | - | Terminated(Business Reasons) | - | - |
| NCT02244242 | Prostatic Hyperplasia | Phase 4 | Completed | - | - |
| NCT01953887 | EC905
Pharmac... more >>okinetics
Healthy Subjects Collapse << | Phase 1 | Completed | - | United Kingdom
... more >> Parexel
London, United Kingdom, HA1 3UJ Collapse << |
| NCT02239484 | Erectile Dysfunction
... more >> Benign Prostatic Hyperplasia Collapse << | Phase 1 | Completed | - | Korea, Republic of
... more >>
Samgsung Seoul Hospital
Seoul, Korea, Republic of Collapse << |
| NCT00448123 | Kidney Stones
... more >> Ureteral Stones Collapse << | Not Applicable | Completed | - | United States, Michigan
... more >>
William Beaumont Hospital
Royal Oak, Michigan, United States, 48073 Collapse << |
| NCT00382265 | Ureterolithiases | Phase 4 | Completed | - | United States, Alabama
... more >>
University of Alabama - Birmingham
Birmingham, Alabama, United States, 35249
United States, District of Columbia
The George Washington University Medical Center
Washington, District of Columbia, United States, 20037
United States, Pennsylvania
Thomas Jefferson University
Philadelphia, Pennsylvania, United States, 19107
University of Pittsburgh
Pittsburgh, Pennsylvania, United States, 15261 Collapse << |
| NCT01534351 | Benign Prostatic Hyperplasia | Phase 3 | Terminated(Business Reasons) | - | - |
| NCT00382265 | - | - | Completed | - | - |
| NCT00954889 | Benign Prostatic Hyperplasia | Not Applicable | Unknown | - | Korea, Republic of
... more >>
Department of Urology, Samsung Medical Center
Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT02972268 | Lower Urinary Tract Symptoms
... more >> Benign Prostatic Hyperplasia Collapse << | Phase 3 | Unknown | December 2017 | Korea, Republic of
... more >>
Asan Medical Center
Recruiting
Seoul, Korea, Republic of
Contact: Myung Soo Choo, MD., Ph.D Collapse << |
| NCT01149733 | Healthy | Phase 1 | Completed | - | Canada, Ontario
... more >>
Pharma Medica Research Inc.
Toronto, Ontario, Canada, M1R 5A3 Collapse << |
| NCT01149733 | - | - | Completed | - | - |
| NCT01149746 | Healthy | Phase 1 | Completed | - | Canada, Ontario
... more >>
Pharma Medica Research Inc.
Toronto, Ontario, Canada, M1R 5A3 Collapse << |
| NCT02827578 | Benign Prostate Hyperplasia | Phase 3 | Unknown | December 2017 | Korea, Republic of
... more >>
Samsung medical center
Recruiting
Seoul, Korea, Republic of
Principal Investigator: Kyu Sung Lee Collapse << |
| NCT00448123 | - | - | Completed | - | - |
| NCT01589263 | Benign Prostatic Hyperplasia
... more >> Lower Urinary Track Symptoms Collapse << | Phase 2 | Recruiting | September 30, 2019 | United States, Texas
... more >>
Michael E. DeBakey VA Medical Center, Houston, TX
Recruiting
Houston, Texas, United States, 77030
Contact: Christopher P Smith, MD 713-791-1414 ChristopherP.Smith@va.gov
Principal Investigator: Christopher P Smith, MD Collapse << |
| NCT02058576 | Urologic Diseases | Phase 1 | Completed | - | United Kingdom
... more >> GSK Investigational Site
Belfast, United Kingdom, BT9 6AD Collapse << |
| NCT01471678 | Prostatic Hyperplasia | Phase 1 | Completed | - | Australia, New South Wales
... more >>
GSK Investigational Site
Randwick, New South Wales, Australia, 2031 Collapse << |
| NCT01673490 | - | - | Terminated(The sole investigat... more >>ive site refused to accept the amended protocol and declined to continue the study. There was no safety signal nor any other reason.) Collapse << | - | - |
| NCT02052713 | Urologic Diseases | Phase 1 | Completed | - | United Kingdom
... more >> GSK Investigational Site
Belfast, United Kingdom, BT9 6AD Collapse << |
| NCT01829893 | Benign Prostatic Hyperplasia (... more >>BPH) Collapse << | Phase 1 | Completed | - | Korea, Republic of
... more >>
Clinical trial center of Anam Hospital
Seoul, Korea, Republic of, 136-705 Collapse << |
| NCT02169713 | Healthy Subjects
... more >> Pharmacokinetics
Drug-Drug Interaction (DDI) Collapse << | Phase 1 | Completed | - | Germany
... more >> Parexel International GmbH
Berlin, Germany, 14050 Collapse << |
| NCT01404637 | Benign Prostatic Hyperplasia | Not Applicable | Unknown | October 2013 | - |
| NCT01254071 | Prostatic Hyperplasia | Phase 1 | Completed | - | Australia, New South Wales
... more >>
GSK Investigational Site
Randwick, New South Wales, Australia, 2031 Collapse << |
| NCT00701779 | Benign Prostatic Hyperplasia | Phase 4 | Completed | - | United States, Indiana
... more >>
Research Institute of Deaconess Clinic
Evansville, Indiana, United States, 47713 Collapse << |
| NCT00701779 | - | - | Completed | - | - |
| NCT01673490 | Prostatic Hyperplasia | Phase 4 | Terminated(The sole investigat... more >>ive site refused to accept the amended protocol and declined to continue the study. There was no safety signal nor any other reason.) Collapse << | - | Vietnam
... more >> GSK Investigational Site
Ho Chi Minh, Vietnam Collapse << |
| NCT02264184 | Healthy | Phase 1 | Completed | - | - |
| NCT02264171 | Healthy | Phase 1 | Completed | - | - |
| NCT02369744 | Ureteral Calculus
... more >> Ureterolithiasis
Ureteral Stone Collapse << | Phase 4 | Terminated(due to lack of suit... more >>able patient population) Collapse << | - | United States, Pennsylvania
... more >>
Einstein Medical Center Philadelphia
Philadelphia, Pennsylvania, United States, 19141 Collapse << |
| NCT02138773 | Healthy
Pharm... more >>acokinetics of Tamsulosin Hydrochloride Collapse << | Phase 1 | Completed | - | China
... more >>
Shanghai, China Collapse << |
| NCT02509104 | Prostatic Hyperplasia | Phase 1 | Completed | - | United States, Maryland
... more >>
GSK Investigational Site
Baltimore, Maryland, United States, 21225 Collapse << |
| NCT02518971 | Postoperative Urinary Retentio... more >>n Collapse << | Phase 3 | Recruiting | December 2019 | United States, Michigan
... more >>
University of Michigan Health System
Recruiting
Ann Arbor, Michigan, United States, 48109
Contact: Aidin Eslam Pour, MD 734-936-5780 aeslampo@med.umich.edu Collapse << |
| NCT02958878 | Postoperative Urinary Retentio... more >>n Collapse << | Phase 4 | Enrolling by invitation | June 2019 | - |
| NCT03524339 | Urinary Retention
... more >> Lower Urinary Tract Symptoms
Stress Urinary Incontinence
Pelvic Organ Prolapse Collapse << | Phase 2
Phase 3 | Recruiting | December 31, 2019 | United States, Ohio
... more >>
University Hospitals Cleveland Medical Center
Recruiting
Cleveland, Ohio, United States, 44106
Contact: Graham Chapman, MD 440-915-5543 graham.chapman@uhhospitals.org
University Hospitals
Recruiting
Cleveland, Ohio, United States, 44106
Contact: Graham C Chapman, MD 440-915-5543 graham.chapman@uhhospitals.org Collapse << |
| NCT03614052 | Ureterolithiasis
... more >> Ureteral Calculi
Urolithiasis Collapse << | Phase 4 | Recruiting | November 30, 2019 | Chile
... more >> Clinica Santa Maria
Recruiting
Santiago, Metropolitana, Chile
Contact: Jose Salvado, MD jasalvadob@gmail.com
Clinica Tabancura
Recruiting
Santiago, Metropolitana, Chile
Contact: Alfredo Domenetch, MD domenech65@gmail.com
Hospital Clinico Pontificia Universidad católica de Chile
Recruiting
Santiago, Metropolitana, Chile
Contact: Gaston Astroza, MD +56223543468 gaeulufi@gmail.com
Hospital La Unión
Recruiting
La Union, Chile
Contact: Matias Westendarp, MD m.westendarp@yahoo.com
Clinica Alemana Valdivia
Recruiting
Valdivia, Chile
Contact: Raul Cañoles, MD rcanoles@hotmail.com Collapse << |
| NCT03709992 | Ureter Stone
... more >>Quality of Life
Ureter Obstruction Collapse << | Not Applicable | Not yet recruiting | November 1, 2019 | - |
| NCT03314025 | Urinary Retention | Phase 2 | Recruiting | October 1, 2019 | Canada, British Columbia
... more >>
St. Paul's Hospital
Not yet recruiting
Vancouver, British Columbia, Canada, V6Z 1Y6
Contact: Rohan Kakkar (604) 682-2344 ext 66040 rkakkar@providencehealth.bc.ca
Principal Investigator: Carl Brown, MD FRCSC
Canada, Ontario
Ottawa Hospital Research Institute
Not yet recruiting
Ottawa, Ontario, Canada, K1Y 4E9
Contact: Nancy Laffin, RN BScN 613-798-5555 ext 18422 nlaffin@toh.ca
Principal Investigator: Reilly Musselman, MD FRCSC
Sub-Investigator: Lara Williams, MD FRCSC
Canada, Quebec
CHU de Quebec - Universite Laval
Recruiting
Quebec City, Quebec, Canada, G1L 3L5
Contact: Sebastien Drolet, MD, FRCSC (418)525-4444 ext 54160 Sebastien.Drolet.chx@gmail.com
Contact: Ann Wright, RN (418)525-4444 ext 53887 Ann.Wright@chuq.qc.ca
Principal Investigator: Sebastien Drolet, MD FRCSC
Sub-Investigator: Claudya Morin, MD
Sub-Investigator: Alexandre Bouchard, MD FRCSC
Sub-Investigator: Philippe Bouchard, MD FRCSC
Sub-Investigator: Alexis Turgeon-Fournier, MD MSc FRCPC Collapse << |
| NCT03750656 | Lower Urinary Tract Symptoms | Phase 4 | Recruiting | November 2022 | United States, Kansas
... more >>
University of Kansas Health System
Recruiting
Kansas City, Kansas, United States, 66160
Contact: Alexandra Dahlgren Collapse << |
| NCT03274700 | Ureteric Stone | Early Phase 1 | Not yet recruiting | October 2018 | - |
| NCT02684344 | Post-Operative Urinary Retenti... more >>on Collapse << | Not Applicable | Terminated(Lack of enrollment) | - | United States, New York
... more >>
University of Rochester
Rochester, New York, United States, 14642 Collapse << |
| NCT01957189 | Prostatic Hyperplasia | Phase 1 | Completed | - | China
... more >> GSK Investigational Site
Shanghai, China, 200030 Collapse << |
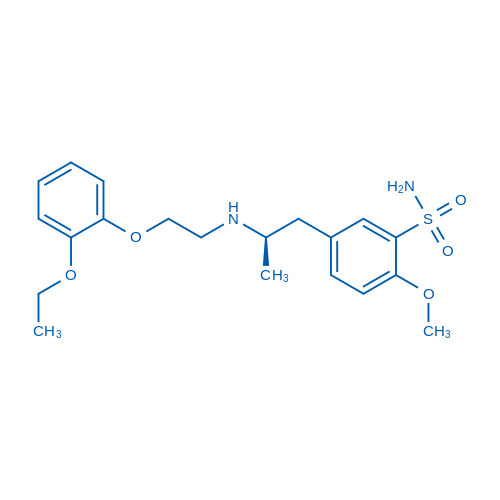

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
