| NCT03419572 | - | - | Recruiting | January 5, 2021 | - |
| NCT00970411 | Solid Tumors | Phase 1 | Completed | - | Japan
... more >>
Shizuoka, Japan Collapse << |
| NCT01058655 | Gastrointestinal Cancer | Phase 1
Phase 2 | Completed | - | United States, Massachusetts
... more >>
Massachusetts General Hospital
Boston, Massachusetts, United States, 02114
Beth Israel Deaconess Medical Center
Boston, Massachusetts, United States, 02115
Dana-Farber Cancer Institute
Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01631097 | Hepatic Impairment | Phase 1 | Completed | - | United States, Florida
... more >>
Miami, Florida, United States
Orlando, Florida, United States Collapse << |
| NCT01058655 | - | - | Completed | - | - |
| NCT01478594 | Colorectal Cancer | Phase 2 | Completed | - | - |
| NCT01030783 | Advanced Renal Cell Carcinoma | Phase 3 | Completed | - | - |
| NCT01076010 | Advanced Renal Cell Carcinoma | Phase 3 | Completed | - | - |
| NCT01835223 | Advanced Adult Hepatocellular ... more >>Carcinoma
Non-Resectable Hepatocellular Carcinoma Collapse << | Phase 1
Phase 2 | Active, not recruiting | December 1, 2019 | United States, New York
... more >>
Roswell Park Cancer Institute
Buffalo, New York, United States, 14263
United States, Ohio
Case Western Reserve University
Cleveland, Ohio, United States, 44106 Collapse << |
| NCT01478594 | - | - | Completed | - | - |
| NCT01745367 | Triple Negative Breast Cancer | Phase 2 | Terminated(low enrollment) | - | - |
| NCT00502307 | Carcinoma, Renal Cell | Phase 2 | Completed | - | India
... more >>
Vellore, Tamil Nadu, India, 632004
Kolkata, India
Mumbai, India
New Delhi, India
Pune, India
Russian Federation
Astrakhan, Russian Federation
Ioshkar-Ola, Russian Federation
Kazan, Russian Federation
Moscow, Russian Federation, 125284
Moscow, Russian Federation, 129128
Moscow, Russian Federation
Novgorod, Russian Federation
Obninsk, Russian Federation
Pyatigorsk, Russian Federation
Rostove-on-Don, Russian Federation
Sochi, Russian Federation
St. Petersburg, Russian Federation
Tomsk, Russian Federation
Ufa, Russian Federation
Ukraine
Cherkassy, Ukraine
Dnepropetrovsk, Ukraine
Donetsk, Ukraine
Kharkov, Ukraine
Lviv, Ukraine
Uzhgorod, Ukraine
Zaporizzhya, Ukraine Collapse << |
| NCT01885949 | Prostate Cancer | Phase 2 | Active, not recruiting | June 2019 | United States, Massachusetts
... more >>
Massachusetts General Hospital
Boston, Massachusetts, United States, 02114 Collapse << |
| NCT01782313 | Recurrent Adult Soft Tissue Sa... more >>rcoma
Stage III Adult Soft Tissue Sarcoma
Stage IV Adult Soft Tissue Sarcoma Collapse << | Phase 2 | Completed | - | United States, Illinois
... more >>
Northwestern University
Chicago, Illinois, United States, 60611
United States, Iowa
University of Iowa
Iowa City, Iowa, United States, 52246
United States, Minnesota
Mayo Clinic
Rochester, Minnesota, United States, 55905
United States, Missouri
Washington University
Saint Louis, Missouri, United States, 63110
United States, Wisconsin
University of Wisconsin
Madison, Wisconsin, United States, 53792 Collapse << |
| NCT01210846 | Advanced Solid Tumors | Phase 1 | Completed | - | United States, Arizona
... more >>
TGEN Clinical Research Service at Scottsdale Healthcare
Scottsdale, Arizona, United States
United States, Florida
Florida Cancer Specialists
Ft. Myers, Florida, United States
United States, Indiana
Horizon Oncology Research, Inc.
Lafayette, Indiana, United States
United States, Louisiana
Jayne Gurtler MD, Laura Brinz MD, Angelo Russo MD and Janet Burroff MD APMC
Metairie, Louisiana, United States
United States, Maryland
Associates in Oncology/Hematology
Rockville, Maryland, United States
United States, Oklahoma
Oklahoma University Cancer Institute (OUCI)
Oklahoma City, Oklahoma, United States
United States, Tennessee
Tennessee Onocology
Nashville, Tennessee, United States
United States, Washington
Multicare Research Institute/Tacoma General Hospital
Tacoma, Washington, United States Collapse << |
| NCT01782313 | - | - | Completed | - | - |
| NCT00660153 | Colorectal Cancer
... more >> Gastrointestinal Cancer Collapse << | Phase 1 | Completed | - | Netherlands
... more >> University Medical Center Groningen; Internal Medicine, Department of Medical Oncology
Groningen, Netherlands
Erasmus Medical Center; Department of Medical Oncology
Rotterdam, Netherlands Collapse << |
| NCT01673386 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Completed | - | - |
| NCT02627963 | Carcinoma, Renal Cell | Phase 3 | Active, not recruiting | December 2019 | - |
| NCT01728181 | Non-small Cell Lung Cancer
... more >> Stage IV Disease
EGFR Unknown or Wild-type Collapse << | Phase 1
Phase 2 | Withdrawn(Stopped before appro... more >>val due to ineffective drug) Collapse << | March 2016 | - |
| NCT01369433 | Solid Tumors | Not Applicable | Completed | - | - |
| NCT00563147 | Renal Cell Carcinoma | Phase 1 | Completed | - | United States, California
... more >>
UCLA Jonsson Comprehensive Cancer Center
Los Angeles, California, United States, 90095
Stanford University
Stanford, California, United States, 94305
United States, Florida
H. Lee Moffitt Cancer Center
Tampa, Florida, United States, 33612
United States, Nebraska
Nebraska Methodist Hospital
Omaha, Nebraska, United States, 68114
United States, Texas
The Methodist Hospital Research Institute
Houston, Texas, United States, 77030 Collapse << |
| NCT01297244 | Renal Cell Carcinoma | Phase 2 | Completed | - | - |
| NCT00717340 | Metastatic Breast Cancer | Phase 1 | Completed | - | United States, Massachusetts
... more >>
Dana Farber Cancer Institute, Inc.
Boston, Massachusetts, United States, 02115
United States, New York
Memorial Sloan-Kettering Cancer Center
New York, New York, United States, 10021
Germany
Universitaetsklinikum Essen
Essen, Germany, D-45147 Collapse << |
| NCT00826878 | Carcinoma, Non-Small-Cell Lung | Phase 1 | Completed | - | United States, District of Col... more >>umbia
Georgetown University
Washington, District of Columbia, United States, 20007
United States, Kansas
Kansas University Medical Center
Kansas City, Kansas, United States, 66160
United States, New York
Memorial Sloan-Kettering
New York, New York, United States, 10021 Collapse << |
| NCT01846871 | Glioblastoma | Phase 2 | Completed | - | United States, Massachusetts
... more >>
Massachusetts General Hospital
Boston, Massachusetts, United States, 02114 Collapse << |
| NCT01306630 | Advanced Solid Tumors
... more >> Locally Advanced or Metastatic Breast or Colorectal Cancer Collapse << | Phase 1 | Completed | - | United States, Arizona
... more >>
Scottsdale, Arizona, United States, 85258
United States, Florida
Fort Myers, Florida, United States, 33908
United States, Tennessee
Nashville, Tennessee, United States, 37203 Collapse << |
| NCT01769885 | Stage II Renal Cell Cancer
... more >> Stage III Renal Cell Cancer Collapse << | Not Applicable | Withdrawn | - | - |
| NCT01363778 | Healthy | Phase 1 | Completed | - | United States, Wisconsin
... more >>
Covance Clinical Research Unit, Inc.
Madison, Wisconsin, United States, 53704 Collapse << |
| NCT01972516 | Ovarian Cancer
... more >> Fallopian Tube Cancer
Primary Peritoneal Carcinoma Collapse << | Phase 2 | Terminated(due to slow accrual... more >>) Collapse << | - | United States, Massachusetts
... more >>
Dana-Farber Cancer Institute
Boston, Massachusetts, United States, 02215 Collapse << |
| NCT01972516 | - | - | Terminated(due to slow accrual... more >>) Collapse << | - | - |
| NCT01853644 | Recurrent Epithelial Ovarian C... more >>ancer
Recurrent Fallopian Tube Cancer
Recurrent Primary Peritoneal Cancer Collapse << | Phase 2 | Active, not recruiting | October 2019 | United States, Illinois
... more >>
Northwestern University
Chicago, Illinois, United States, 60611
CDH-Delnor Health System - Northwestern Medicine Cancer Center
Warrenville, Illinois, United States, 60555 Collapse << |
| NCT01363804 | Healthy | Phase 1 | Completed | - | United States, Florida
... more >>
Covance Clinical Research Unit, Inc.
Daytona Beach, Florida, United States, 32117 Collapse << |
| NCT01834183 | Renal Cell Carcinoma | Phase 2 | Withdrawn(Sponsor Aveo has wit... more >>hdrawn support for this trial.) Collapse << | - | United States, Massachusetts
... more >>
Massachusetts General Hospital
Boston, Massachusetts, United States, 02114
Beth Israel Deaconess Medical Center
Boston, Massachusetts, United States, 02215
Brigham and Women's Hospital
Boston, Massachusetts, United States, 02215
Dana-Farber Cancer Institute
Boston, Massachusetts, United States, 02215 Collapse << |
| NCT01807156 | - | - | Terminated(Primary endpoint no... more >>t met) Collapse << | - | - |
| NCT01316848 | Food Effect of Tivozanib in He... more >>alth Subjects Collapse << | Phase 1 | Completed | - | United States, Texas
... more >>
Covance Clinical Research Unit, Inc.
Dallas, Texas, United States Collapse << |
| NCT01807156 | Hepatocellular Cancer | Phase 2 | Terminated(Primary endpoint no... more >>t met) Collapse << | - | United States, Georgia
... more >>
Emory University, Winship Cancer Institute
Atlanta, Georgia, United States, 30322 Collapse << |
| NCT03136627 | Carcinoma, Renal Cell | Phase 1
Phase 2 | Active, not recruiting | December 2018 | France
... more >> Bordeaux Hospital University Center (CHU)
Bordeaux, France
Center Léon Bérard
Lyon, France
Institut Paoli-Calmettes
Marseille, France
Centre Paul Strauss
Strasbourg, France, 67065
Institut Gustave Roussy
Villejuif, France, 94805 Collapse << |
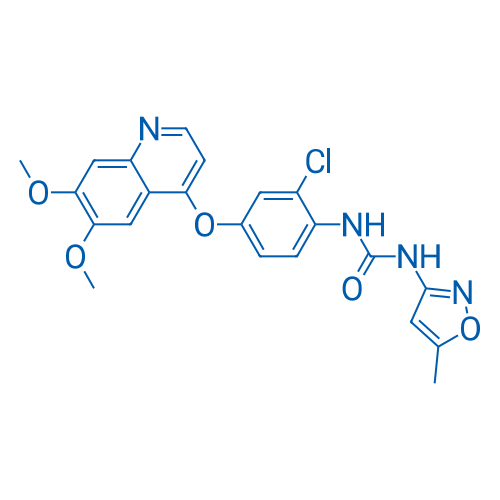

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
