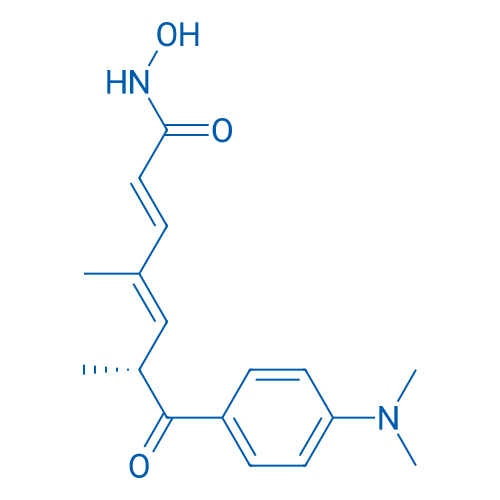
CAS No.: 58880-19-6
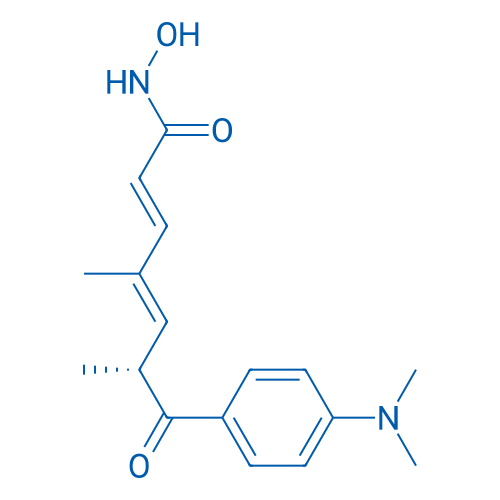
Trichostatin A/曲古抑菌素A Catalog No. CSN12139
Synonyms: TSA;曲古抑菌素A
Trichostatin A is a selective and potent HDAC inhibitor with IC50 of ~1.8 nM and HDAC8 is the only known member of the HDAC-family that is not affected by TSA.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 HDAC
IC50:~1.8nM- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT03049124 Rectal Cancer Not Applicable Recruiting July 31, 2019 Canada, Ontario ... more >> University of Ottawa Recruiting Ottawa, Ontario, Canada, K1N 6N5 Contact: Amanda Wurz, MSc 613-562-5800 ext 3626 amanda.wurz@uottawa.ca Collapse << NCT02152267 Craniomandibular Disorders Not Applicable Completed - Brazil ... more >> Federal University of Bahia Salvador, Bahia, Brazil, 40110-902 Collapse << NCT01028014 Urethral Sphincter Activity Not Applicable Completed - United States, Alabama ... more >> University of Alabama at Birmingham, The Kirklin Clinic Birmingham, Alabama, United States, 35233 Collapse << - 更多
- 参考文献
- [1] Yoon S, Eom GH, et al. HDAC and HDAC Inhibitor: From Cancer to Cardiovascular Diseases. Chonnam Med J. 2016 Jan;52(1):1-11.
- [2] Vigushin DM, Ali S, et al. Trichostatin A is a histone deacetylase inhibitor with potent antitumor activity against breast cancer in vivo. Clin Cancer Res. 2001 Apr;7(4):971-6.
- [3] Xu WS, Parmigiani RB, et al. Histone deacetylase inhibitors: molecular mechanisms of action. Oncogene. 2007 Aug 13;26(37):5541-52.
- [4] Codd R, Braich N, et al. Zn(II)-dependent histone deacetylase inhibitors: suberoylanilide hydroxamic acid and trichostatin A. Int J Biochem Cell Biol. 2009 Apr;41(4):736-9.
- [5] Sanderson L, Taylor GW, et al. Plasma pharmacokinetics and metabolism of the histone deacetylase inhibitor trichostatin a after intraperitoneal administration to mice. Drug Metab Dispos. 2004 Oct;32(10):1132-8. Epub 2004 Jul 21.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 58880-19-6 | 储存条件 |
|
|||||||||
| 分子式 | C17H22N2O3 | 运输 | 蓝冰 | |||||||||
| 分子量 | 302.37 | 别名 | TSA;曲古抑菌素A | |||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| A2780 | 0.05-2 μM | Growth Inhibition Assay | 10/24/48 h | mediates growth arrest in a concentration- and time-dependent manner | 24223801 |
| A431 | 2/10/50/100nM | Apoptosis Assay | 48h | inhibits the cell growth | 25371069 |
| A431 | 50nM | Function Assay | 2/6/12/24h | activates p21 and inhibits ATF3 expression | 25371069 |
| A549 | 1-10μM | Growth Inhibition Assay | 24h | IC50 of 3.2μM | 25169491 |
| A549 | 0.1-1μM | Growth Inhibition Assay | 48h | IC50 of 0.28μM | 25169491 |
| A549 | 0.01-0.9μM | Growth Inhibition Assay | 72h | IC50 of 0.06μM | 25169491 |
| A549 | 0.25/0.5/1 μM | Growth Inhibition Assay | 24/48/72 h | inhibits the cell growth in concentration and time-dependent manner | 23867991 |
| A549 | 0.5/1 μM | Apoptosis Assay | 48 h | induces cell apoptosis | 23867991 |
| A549 | 250 nM | Growth Inhibition Assay | 6-72 h | causes a greater inhibitory effect combine with TXT or erlotinib | 22994780 |
| ACP02 | 250/350/500nM | Growth Inhibition Assay | 24h | decreases cell viability of approximately 70 % at 250 nM | 24668547 |
| ACP03 | 250/350/500nM | Growth Inhibition Assay | 24h | decreases cell viability of approximately 70 % at 250 nM | 24668547 |
| AGS | 0.015-1 μM | Growth Inhibition Assay | 72 h | induces cell death concentration dependently | 23745024 |
| ARN8 | 0.05-2 μM | Function Assay | 24 h | reduces the induction of p53-dependent genes by Nutlin-3 | 23470540 |
| CA46 | 3/6/12/24/48 nM | Growth Inhibition Assay | 48 h | inhibits the cell proliferation at 24 and 48 nM | 24064951 |
| Caco-2 | 1 µM | Function Assay | 24 h | decreases SERT protein expression | 23195070 |
| CNE2 | 100-600nM | Growth Inhibition Assay | 24/48/72h | inhibits the proliferation in a time- and dose-dependent manner | 24969901 |
| ECC1 | 500 nM | Function Assay | 5 d | induces L1CAM expression co-treatment with 5-AzaC | 23530769 |
| ECC-1 | 100 nM | Growth Inhibition Assay | 24 h | increases the proportion of apoptotic nuclei to 35% | 23028803 |
| EN1 | 500 nM | Function Assay | 5 d | induces L1CAM expression co-treatment with 5-AzaC | 23530769 |
| H1299 | 1 μM | Growth Inhibition Assay | 24/48/72 h | increases cell death after 24h | 23916609 |
| H1299 | 0.25/0.5/1 μM | Growth Inhibition Assay | 24/48/72 h | inhibits the cell growth in concentration and time-dependent manner | 23867991 |
| H1299 | 0.5/1 μM | Apoptosis Assay | 48 h | induces cell apoptosis | 23867991 |
| H1299 | 0.33–1 µM | Function Assay | 24/48 h | increases E-cadherin protein levels dose dependently | 23461975 |
| H1299 | 0.5 μM | Function Assay | 48 h | inhibits the migratory potential combine with silibinin | 23461975 |
| H1299 | 0.5 μM | Function Assay | 48 h | inhibits the invasiveness combine with silibinin | 23461975 |
| HASMCs | 0-500 nM | Function Assay | 6 d | down-regulates the expression of HADC and cell viability | 23518467 |
| HCT1 | 0.2/1.0/5.0 μM | Growth Inhibition Assay | 12/24/36/48 h | induces cell death in concentration- and time-dependent manner | 23770000 |
| HCT116 | 0.2 μM | Growth Inhibition Assay | 12 h | enhances cell apoptosis induced by radiation | 24122231 |
| HEC1A | 500 nM | Function Assay | 5 d | induces L1CAM expression co-treatment with 5-AzaC | 23530769 |
| HEC-1A | 100 nM | Growth Inhibition Assay | 24 h | increases the proportion of apoptotic nuclei to 39% | 23028803 |
| HEK 293 | 0.7μM | Function Assay | 24h | enhances ENaC acetylation and increases ENaC abundance in the total cell lysate and at the cell surface | 25787079 |
| HEK293 | 1 µM | Function Assay | 18 h | inhibits the kalirin-7-mediated recruitment of synphilin-1 aggregates into aggresomes | 23284848 |
| HEL | 0.5 μM | Function Assay | 24 h | decreases the enzyme activity of HDACs co-treatment with curcumin | 23430957 |
| HeLa | 250nM | Function Assay | 16h | increases CYP1A1 mRNA expression | 25116688 |
| HeLa | 2μM | Growth Inhibition Assay | 48h | decreases cell viability about 25% | 24846135 |
| HeLa | 1mM | Function Assay | 0.5h | abolishes the total HDAC activity | 24707474 |
| HeLa | 100 nM | Growth Inhibition Assay | 24 h | IC50 of 100nM | 23165748 |
| HeLa | 40 nM | Growth Inhibition Assay | 48 h | IC50 of 40nM | 23165748 |
| HeLa | 20 nM | Growth Inhibition Assay | 72 h | IC50 of 20nM | 23165748 |
| HeLa | 10/30/50 nM | Growth Inhibition Assay | 72 h | increases the number of MMP (ΔΨm) loss cells dose dependently | 23165748 |
| HL60 | 1 μM | Apoptosis Assay | 24 h | induces cell death | 23400519 |
| HL60 | 150-350 nM | Growth Inhibition Assay | 24 h | increases cell apoptosis when concentrations higher than 250 nM | 22753739 |
| HLEB3 | 0.2 μM | Growth Inhibition Assay | 48 h | suppresses cell proliferation through suppressing the PI3K/Akt, p38MAPK and ERK1/2 signaling pathways | 24157878 |
| HLEB3 | 0.4/0.8 μM | Growth Inhibition Assay | 48 h | induces cell apoptosis | 24157878 |
| HMEC-1 | 300nM | Function Assay | 24h | increases expression of VEGFR3 mRNA | 24710631 |
| hMSCs | 6.25 nM | Function Assay | 24 h | stabilizes Histone Acetylation and the Expression of Pluripotent Genes | 24312356 |
| HT29 | 180 nM | Growth Inhibition Assay | 72 h | IC50 of 180 nM | 24368265 |
| HTK | 400 nM | Function Assay | 72 h | inhibits TGF-β–Induced Myofibroblast Differentiation | 23284002 |
| HTK | 100-800nM | Function Assay | 72 h | blocks TGF-β–Induced ROS and H2O2 Accumulation | 23284002 |
| Huh7 | 0.1/0.5/1.0 μM | Function Assay | 24h | suppresses YTHDC2 gene expression | 24269672 |
| Huh7 | 1 μM | Growth Inhibition Assay | 24 h | reduces about 20% viability | 23643933 |
| K562 | 0.5 μM | Function Assay | 24 h | decreases the enzyme activity of HDACs co-treatment with curcumin | 23430957 |
| K562 | 1 μM | Apoptosis Assay | 24 h | induces cell death | 23400519 |
| K562 | 1 μM | Function Assay | 12 h | enhances the expression of RUNX3 induced by 5-aza-CdR | 22179198 |
| Kazumi | 1 μM | Apoptosis Assay | 24 h | induces cell death | 23400519 |
| KG1 | 1 μM | Apoptosis Assay | 24 h | induces cell death | 23400519 |
| LM8 | 0.3 μM | Growth Inhibition Assay | 12-96 h | inhibits the cell growth co-treatment with metformin | 23451817 |
| LNCaP | 100-1000nM | Growth Inhibition Assay | 24h | IC50 of 300nM | 24854658 |
| Lovo | 0.2/1.0/5.0 μM | Growth Inhibition Assay | 12/24/36/48 h | induces cell death in concentration- and time-dependent manner | 23770000 |
| MCF7 | 0-600nM | Growth Inhibition Assay | 24h | IC50 of 75nM | 25192721 |
| MCF7 | 0.05-2 μM | Function Assay | 24 h | reduces the induction of p53-dependent genes by Nutlin-3 | 23470540 |
| MCF-7 | 25-400 nM | Growth Inhibition Assay | 48 h | IC50 of 220.4nM | 23055198 |
| MDA-MB-231 | 0-600nM | Growth Inhibition Assay | 24h | IC50 of 100nM | 25192721 |
| MDA-MB-231 | 25-400 nM | Growth Inhibition Assay | 48 h | IC50 of 263.2nM | 23055198 |
| MEFs | 5μM | Function Assay | 16h | increases the EPEC attachment, Tir delivery and the efficiency of pedestal formation | 25482634 |
| MFE296 | 500 nM | Function Assay | 5 d | induces L1CAM expression co-treatment with 5-AzaC | 23530769 |
| MG-63 | 0.3 μM | Growth Inhibition Assay | 12-96 h | inhibits the cell growth co-treatment with metformin | 23451817 |
| MG-63 | 300 nM | Growth Inhibition Assay | 12 h | inhibits the cell growth to 86% | 22799338 |
| MG-63 | 300 nM | Growth Inhibition Assay | 24 h | inhibits the cell growth to 67% | 22799338 |
| MG-63 | 300 nM | Growth Inhibition Assay | 48 h | inhibits the cell growth to 56% | 22799338 |
| NHAC-kn | 10/100/500 nM | Growth Inhibition Assay | 12 h | IC50 of 500nM | 23017871 |
| PC3 | 0.1μM | Function Assay | 48h | reverses EMT | 25434997 |
| PC3 | 0.1μM | Function Assay | 48h | attenuates invasion and migration | 25434997 |
| PC3 | 0.1μM | Function Assay | 48h | induces increase of HDAC1 and HDAC2 on Slug genes promoter | 25434997 |
| PC3 | 100-1000nM | Growth Inhibition Assay | 24h | IC50 of 300nM | 24854658 |
| PMNs | 30 nM | Function Assay | 48 h | inhibits efferocytosis, HDAC activity and Rac activity | 23988617 |
| Reh | 0.3/1 μM | Function Assay | 12 h | enhances the expression of RUNX3 induced by 5-aza-CdR | 22179198 |
| RPE | 0.2/0.4/0.8/1 μM | Growth Inhibition Assay | 24/48/72 h | inhibits the proliferation by cell cycle arrest | 24456602 |
| SCC-6 | 200-3200 nM | Growth Inhibition Assay | 12/24/48 h | inhibits the proliferation of SCC-6 cells in a dose- and time-dependent manner | 22552321 |
| SH-SY5Y | 250 nM | Function Assay | 16 h | changes expression pattern of genes involved in cholesterol synthesis, uptake and efflux | 23326422 |
| SKOV3 | 0.05-2 μM | Growth Inhibition Assay | 10/24/48 h | mediates growth arrest in a concentration- and time-dependent manner | 24223801 |
| SKOV-3 | 1-10μM | Growth Inhibition Assay | 24h | IC50 of 5.6μM | 25169491 |
| SKOV-3 | 0.1-1μM | Growth Inhibition Assay | 48h | IC50 of 0.7μM | 25169491 |
| SKOV-3 | 0.01-0.9μM | Growth Inhibition Assay | 72h | IC50 of 0.32μM | 25169491 |
| SRA01/04 | 0.2 μM | Growth Inhibition Assay | 48 h | suppresses cell proliferation through suppressing the PI3K/Akt, p38MAPK and ERK1/2 signaling pathways | 24157878 |
| SRA01/04 | 0.4/0.8 μM | Growth Inhibition Assay | 48 h | induces cell apoptosis | 24157878 |
| SUM149PT | 2/7.5/10 μM | Growth Inhibition Assay | 48 h | induces cell death at 2 μM insensitively | 23792638 |
| SUM190PT | 50/100/250 nM | Growth Inhibition Assay | 48 h | induces cell death at 50 nM sensitively | 23792638 |
| SW480 | 0.1μM | Function Assay | 48h | reverses EMT | 25434997 |
| SW480 | 0.1μM | Function Assay | 48h | attenuates invasion and migration | 25434997 |
| SW480 | 0.1μM | Function Assay | 48h | induces increase of HDAC1 and HDAC2 on Slug genes promoter | 25434997 |
| TE13 | 0.3μM | Function Assay | 24h | up-regulates RASSF5A level | 25579665 |
| TE13 | 0.3μM | Apoptosis Assay | 24h | inhibits the cell proliferation | 25579665 |
| THP1 | 1 μM | Apoptosis Assay | 24 h | induces cell death | 23400519 |
| U373 | 0.25/0.5/1 μM | Function Assay | 24 h | increases PR-B expression co-treatment with 5AzadC | 23474171 |
| U87 | 100-300 ng | Growth Inhibition Assay | 24 h | inhibits the cell growth to 72% at 200ng | 22270849 |
| U87 GBM | 100/300/500 nM | Growth Inhibition Assay | 72 h | reduces mean cell number by 31, 54, and 58 % | 24464841 |
| U87 GBM | 100/500 Nm | Function Assay | 48 h | Induces Senescence-Like Alterations in Nuclear Morphology | 24464841 |
| U937 | 150-350 nM | Growth Inhibition Assay | 24 h | increases cell apoptosis when concentrations higher than 250 nM | 22753739 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT03049124 | Rectal Cancer | Not Applicable | Recruiting | July 31, 2019 | Canada, Ontario ... more >> University of Ottawa Recruiting Ottawa, Ontario, Canada, K1N 6N5 Contact: Amanda Wurz, MSc 613-562-5800 ext 3626 amanda.wurz@uottawa.ca Collapse << |
| NCT02152267 | Craniomandibular Disorders | Not Applicable | Completed | - | Brazil ... more >> Federal University of Bahia Salvador, Bahia, Brazil, 40110-902 Collapse << |
| NCT01028014 | Urethral Sphincter Activity | Not Applicable | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham, The Kirklin Clinic Birmingham, Alabama, United States, 35233 Collapse << |
| NCT01028014 | - | - | Completed | - | - |
| NCT03016728 | Adolescent and Young Adult Can... more >>cer Survivors Collapse << | Not Applicable | Recruiting | February 2019 | Canada, Ontario ... more >> University of Ottawa Recruiting Ottawa, Ontario, Canada, K1N 6N5 Contact: Amanda Wurz, MSc 613-562-5800 ext 3626 amanda.wurz@uottawa.ca Collapse << |
| NCT01974726 | Eustachian Tube Dysfunction | Not Applicable | Recruiting | July 2019 | United States, Pennsylvania ... more >> University of Pittsburgh Middle Ear Physiology Laboratory Recruiting Pittsburgh, Pennsylvania, United States, 15213 Sub-Investigator: Cuneyt M Alper, MD Sub-Investigator: Margaretha L Casselbrant, MD, PhD Sub-Investigator: Charles D Bluestone, MD Sub-Investigator: Ellen M Mandel, MD Sub-Investigator: Douglas J Swarts, PhD Sub-Investigator: Miriam S Teixeira, MD Collapse << |
| NCT03561688 | Rheumatoid Arthritis ... more >> Rheumatoid Arthritis of Foot (Disorder) Collapse << | Not Applicable | Recruiting | April 1, 2020 | Denmark ... more >> King Christian X's Hospital for Rhuematic Diseases Recruiting Gråsten, Denmark, 6300 Contact: Kim Hørslev-Petersen, MD, Phd Department for Rheumatology Recruiting Hjørring, Denmark, 9800 Contact: Marcin R Kowalski, MD, Phd Collapse << |
| NCT01853215 | Intraosseous Vascular Access | Not Applicable | Completed | - | United States, Texas ... more >> Bulverde-Spring Branch EMS Spring Branch, Texas, United States, 78070 Collapse << |
| NCT01898208 | - | - | Completed | - | - |
| NCT01853215 | - | - | Completed | - | - |
| NCT00168454 | Overactive Bladder ... more >> Urinary Incontinence Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Pittsburgh, Pennsylvania, United States Belgium Ghent, Belgium Canada, British Columbia Victoria, British Columbia, Canada Germany Berlin, Germany Poland Warsaw, Poland United Kingdom Sheffield, United Kingdom Collapse << |
| NCT01898208 | Bacteremia Fu... more >>ngemia Bloodstream Infection Collapse << | Not Applicable | Completed | - | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00168454 | - | - | Completed | - | - |
| NCT00499460 | - | - | Completed | - | - |
| NCT01332513 | Epilepsy | Phase 1 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT03636035 | Mild Knee Osteoarthritis | Not Applicable | Not yet recruiting | December 31, 2019 | - |
| NCT02013375 | Sickle Cell Disease | Phase 2 | Active, not recruiting | October 1, 2018 | United States, Illinois ... more >> University of Illinois Cancer Center Chicago, Illinois, United States, 60612 Collapse << |
| NCT00499460 | Healthy, No Evidence of Diseas... more >>e Collapse << | Phase 4 | Completed | - | United States, Washington ... more >> Fred Hutchinson Cancer Research Center Seattle, Washington, United States, 98109-1024 Collapse << |
| NCT02638493 | - | - | Completed | - | - |
| NCT03490162 | Malaria | Phase 1 | Recruiting | June 4, 2019 | United States, North Carolina ... more >> Duke University School of Medicine - Duke Clinical Research Institute - Duke Clinical Research Unit Recruiting Durham, North Carolina, United States, 27710 Collapse << |
| NCT00916721 | Smoking Cessation | Phase 3 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital - Center For Addiction Medicine Boston, Massachusetts, United States, 02114 Collapse << |
| NCT03514316 | Orthodontic Appliance Complica... more >>tion Gingival Overgrowth Collapse << | Not Applicable | Completed | - | Italy ... more >> Department of Orthodontics, Department of Clinical Sciences and Translational Medicine, University of Rome "Tor Vergata". Roma, Italy, 00133 Collapse << |
| NCT02638493 | - | - | Completed | - | United States, North Carolina ... more >> University of North Carolina Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT00916721 | - | - | Completed | - | - |
| NCT01582503 | Asthma | Phase 2 | Completed | - | - |
| NCT00311376 | Overactive Bladder | Phase 3 | Completed | - | United States, Michigan ... more >> Royal Oak, Michigan, United States Australia, New South Wales Randwick, New South Wales, Australia Austria Innsbruck, Austria Belgium Gent, Belgium Canada Sherbrooke, Canada Czech Republic Ostrava, Czech Republic France Garches, France Germany Halle (Saale), Germany New Zealand Christchurch, New Zealand Poland Wroclaw, Poland Russian Federation Moscow, Russian Federation Slovakia Martin, Slovakia Ukraine Lviv, Ukraine United Kingdom London, United Kingdom Collapse << |
| NCT00096850 | HIV Infections ... more >> Tuberculosis Collapse << | Not Applicable | Completed | - | United States, California ... more >> Stanford CRS Palo Alto, California, United States, 94305-5107 United States, Ohio The Ohio State Univ. AIDS CRS Columbus, Ohio, United States, 43210 United States, Tennessee Vanderbilt Therapeutics CRS Nashville, Tennessee, United States, 37203 Collapse << |
| NCT00461292 | Overactive Bladder | Phase 3 | Completed | - | United States, Connecticut ... more >> Middlebury, Connecticut, United States Brazil Rio de Janeiro, Brazil Canada, British Columbia Victoria, British Columbia, Canada France Salouel, France Italy Milan, Italy Netherlands Amsterdam, Netherlands Portugal Porto, Portugal Singapore Singapore, Singapore South Africa Pretoria, South Africa Spain Tenerife, Spain Taiwan Hualien, Taiwan United Kingdom Scunthorpe, United Kingdom Collapse << |
| NCT01865760 | Obesity Hypog... more >>lycemia Surgery Collapse << | Not Applicable | Unknown | March 2016 | Denmark ... more >> Department of Endocrinology, Aarhus University Hospital Recruiting Aarhus C, Denmark, 8000 Contact: Joan Bach Nielsen, Dr 0045+24818454 joanni@rm.dk Sub-Investigator: Joan Bach Nielsen, MD Collapse << |
| NCT00467753 | Autism | Not Applicable | Completed | - | United States, New Jersey ... more >> Child and Adolescent Psychiatry at the University Behavioral Healthcare Building, UMDNJ-RWJMS Piscataway, New Jersey, United States, 08854 Collapse << |
| NCT00035425 | Febrile Neutropenia | Phase 3 | Completed | - | - |
| NCT02706535 | Drug Interactions | Phase 1 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT00311376 | - | - | Completed | - | - |
| NCT00263211 | Breast Neoplasms | Phase 2 | Terminated(Stopped due to low ... more >>percentage of patients with detectable CTCs at baseline.) Collapse << | - | United States, Missouri ... more >> Washington University School of Medicine St. Louis, Missouri, United States, 63110 Collapse << |
| NCT00263211 | - | - | Terminated(Stopped due to low ... more >>percentage of patients with detectable CTCs at baseline.) Collapse << | - | - |
| NCT03604159 | Opioid Dependence | Phase 4 | Not yet recruiting | August 2020 | - |
| NCT02237586 | Plasmodium Falciparum Malaria ... more >> Malaria Collapse << | Phase 1 | Completed | - | Gabon ... more >> Centre de Recherches Médicales de Lambaréné Lambaréné, Gabon, BP 118 Collapse << |
| NCT00461292 | - | - | Completed | - | - |
| NCT03601663 | Physical Activity | Not Applicable | Recruiting | August 2019 | Canada, Ontario ... more >> University of Ottawa Recruiting Ottawa, Ontario, Canada, K1N6N5 Contact: Jennifer Brunet, PhD (613) 562-5800 ext 3068 jennifer.brunet@uottawa.ca Contact: Melissa Black, BA (613) 562-5800 ext 7300 melissa.black@uottawa.ca Collapse << |
| NCT01756404 | Healthy | Phase 1 | Completed | - | United States, Florida ... more >> Miramar, Florida, United States Collapse << |
| NCT01783704 | Hip Fracture | Not Applicable | Completed | - | United States, Connecticut ... more >> University of Connecticut Health Center Farmington, Connecticut, United States, 06030 United States, Maryland University of Maryland, Baltimore Baltimore, Maryland, United States, 21201 United States, New Hampshire Dartmouth Medical School Lebanon, New Hampshire, United States, 03756 United States, Pennsylvania Arcadia University Glenside, Pennsylvania, United States, 19038-3295 Collapse << |
| NCT03135132 | Visceral Fat ... more >>Weight Loss Collapse << | Not Applicable | Completed | - | Korea, Republic of ... more >> Yonsei University Seoul, Korea, Republic of, 03722 Collapse << |
| NCT00467753 | - | - | Completed | - | - |
| NCT02506140 | Stroke Ischem... more >>ic Attack, Transient Collapse << | Phase 2 Phase 3 | Active, not recruiting | March 10, 2018 | China, Fujian ... more >> The First Affiliated Hospital of Fujian Medical University Fuzhou, Fujian, China, 350005 China, Guangdong The Second People's Hospital of Shenzhen Shenzhen, Guangdong, China, 518000 China, Hebei The Second Hospital of Hebei Medical University Shijiazhuang, Hebei, China, 050000 North China University of Science And Technology Affiliated Hospital Tangshan, Hebei, China, 063000 Tangshan Gongren Hospital Tangshan, Hebei, China, 063000 China, Henan The First Affiliated Hospital of Zhengzhou University Zhengzhou, Henan, China, 450052 China, Hubei Wuhan Brain Hospital,General Hospital of The Yangtze River Shipping Wuhan, Hubei, China, 430014 Union Hospital,Tongji Medical College,Huazhong University of Science and Technology Wuhan, Hubei, China, 430022 Wuhan No.1 Hospital Wuhan, Hubei, China, 430022 Renmin Hospital of Wuhan University Wuhan, Hubei, China, 430060 China, Hunan Xiangya Hospital Central South University Changsha, Hunan, China, 410008 China, Jiangsu Northern Jiangsu People's Hospital,Clinical Medical School,YangZhou University Yangzhou, Jiangsu, China, 225001 China, Liaoning General Hospital of Shenyang Military Shengyang, Liaoning, China, 110016 China, Shanxi The Second Hospital of Shanxi Medical University Taiyuan, Shanxi, China, 030001 General Hospital of TISCO Taiyuan, Shanxi, China, 030003 China, Zhejiang Taizhou First People's Hospital,Huangyan Hospital of Wenzhou Medical University Taizhou, Zhejiang, China, 318020 The First Affiliated Hospital of Wenzhou Medical University Wenzhou, Zhejiang, China, 325000 Wenzhou Hospital of integrated Chinese and Western medicine Wenzhou, Zhejiang, China, 325000 China Aviation General Hospital of China Medical University Beijing, China, 100012 Beijing Tian Tan Hospital, Capital Medical University Beijing, China, 100050 Dongfang Hospital Beijing University of Chinese Medicine Beijing, China, 100078 The First Hospital of Fangshan District,Beijing Beijing, China, 102400 Daping Hospital,Third Military Medical University Chongqing, China, 400042 Renji Hospital,Shanghai Jiao Tong University School of Medicine Shanghai, China, 200127 Tianjin Huanhu Hospital Tianjin, China, 300350 Collapse << |
| NCT02869009 | Ischemic Stroke | Early Phase 1 | Recruiting | April 30, 2020 | China, Liaoning ... more >> General Hospital of Shenyang Military Region Recruiting Shenyang, Liaoning, China Contact: Hui-Sheng Chen 86-24-28897511 chszh@aliyun.com Contact: Xin-hong Wang 86-15309885658 450341972@qq.com Collapse << |
| NCT02225886 | Anaemia Response to the Treatm... more >>ent Peripheral Iron Indices Oxalemia Collapse << | Phase 4 | Not yet recruiting | December 2021 | Romania ... more >> "Nefrolab" Dialysis Center Not yet recruiting Slatina, Romania Contact: Aurelian Simionescu, MD, PhD +40732161768 relusimionescu@yahoo.com Contact: Ileana Mihailescu, MD +40730577485 dr_ilemih@yahoo.com Collapse << |
| NCT02523599 | Pain, Postoperative | Phase 3 | Completed | - | United States, Alabama ... more >> Mobile, Alabama, United States, 36617 Sheffield, Alabama, United States, 35660 United States, Arizona Phoenix, Arizona, United States, 85023 United States, Arkansas Little Rock, Arkansas, United States, 72205 United States, California Encino, California, United States, 91436 United States, Florida Bradenton, Florida, United States, 34206 Clermont, Florida, United States, 34711 Inverness, Florida, United States, 34452 Kissimmee, Florida, United States, 34741 Miami, Florida, United States, 33136 Miami, Florida, United States, 33144 Orlando, Florida, United States, 32825 St. Petersberg, Florida, United States, 33713 Tampa, Florida, United States, 33606 Weston, Florida, United States, 33331 United States, Indiana Indianapolis, Indiana, United States, 46202 United States, Massachusetts Springfield, Massachusetts, United States, 01199 United States, Michigan Flint, Michigan, United States, 48532 United States, Nevada Las Vegas, Nevada, United States, 89102 Las Vegas, Nevada, United States, 89106 United States, Texas El Paso, Texas, United States, 79935 Houston, Texas, United States, 77054 Lampasas, Texas, United States, 76550 Collapse << |
| NCT00979589 | Stroke Transi... more >>ent Ischemic Attack Collapse << | Phase 3 | Completed | - | China ... more >> Beijing Tian Tan Hospital, Capital Medical University Beijing, China, 100050 Collapse << |
| NCT02525133 | Pain, Postoperative | Phase 3 | Completed | - | - |
| NCT00817999 | - | - | Completed | - | - |
| NCT02992639 | Visceral Fat ... more >>Liver Dysfunction Collapse << | Not Applicable | Completed | - | Korea, Republic of ... more >> Yonsei University Seoul, Korea, Republic of, 03722 Collapse << |
| NCT01133210 | Hypertriglyceridemia | Phase 1 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine St. Louis, Missouri, United States, 63110 Collapse << |
| NCT00734162 | Hepatitis B Virus (HBV) | Phase 3 | Completed | - | United States, California ... more >> Children's Hospital & Research Center at Oakland Oakland, California, United States, 94609 United States, Indiana Riley Hospital for Children Indianapolis, Indiana, United States, 46202 United States, Washington Children's Hospital & Regional Medical Center, d/b/a Seattle Children's Research Institute Seattle, Washington, United States, 98105 Bulgaria Multiprofile Hospital for Active Treatment Sveti Georgi Plovdiv, Bulgaria, 4002 Clinic of Gastroenterology, Specialized Hospital for Active Treatment of Pediatric Diseases, Sofia Sofia, Bulgaria, 1606 France Hopital Femmes Meres Enfants Bron Cedex, France, 69677 Hôpital Claude Huriez Lille Cedex, France, 59037 Poland Samodzielny Publiczny Dzieciecy Szpital Kliniczny Akademii Medycznej w Bialymstoku Białystok, Poland, 15-274 Wojewodzki Specjalistyczny Szpital im Bieganskiego Bydgoszcz, Poland, 85-030 Wojewodzki Szpital Obserwacyjno-Zakazny im. T. Browicza Bydgoszcz, Poland, 85-030 Krakowski Szpital Specjalistyczny im. Jana Pawla II Kraków, Poland, 31-202 Samodzielny Publiczny Szpital Kliniczny im. Karola Johschera Poznan, Poland, 60-572 Specjalistyczny Zespol Opieki Zdrowotnej nad Matka i Dzieckiem Poznań, Poland, 61-734 Wojewodzki Szpital Zakazny Warszawa, Poland, 01-201 Samodzielny Publiczny Szpital Kliniczny Nr 1 Wrocław, Poland, 50-368 Romania Fundeni Clinical Institute Bucharest, Romania, 022328 Institute for Infectious Diseases Bucharest, Romania, 21105 Cluj Childrens Emergency Hospital Napaco, Romania, 400217 Spain Hosp Univ y Politecnico La Fe de Valencia Madrid, Spain, 28046 Hospital Universitario De Getafe Madrid, Spain, 46009 Turkey Ege Universitesi Tip Fakultesi Hastanesi Izmir, Turkey, 35100 Collapse << |
| NCT02986815 | Glioma | Not Applicable | Recruiting | December 2021 | United States, Texas ... more >> University of Texas Southwestern Medical Center Recruiting Dallas, Texas, United States, 75063 Contact: Sarh McNeil, RN 214-645-8166 sarah.mcneil@utsouthwestern.edu Principal Investigator: Robert Bachoo, MD Collapse << |
| NCT03653546 | Non-small Cell Lung Cancer ... more >> EGFR Gene Mutation Brain Metastases Collapse << | Phase 2 Phase 3 | Recruiting | October 16, 2021 | - |
| NCT00734162 | - | - | Completed | - | - |
| NCT01808040 | Post Menopausal, Hormone Recep... more >>tor Positive Breast Cancer Collapse << | Phase 1 | Completed | - | United States, Wisconsin ... more >> University of Wisconsin Madison, Wisconsin, United States, 53792 Collapse << |
| NCT01663194 | - | - | Completed | - | Turkey ... more >> Erciyes University school of medicine Kayseri, Turkey, 38039 Collapse << |
| NCT02642133 | Ankyloglossia ... more >> Breastfeeding Collapse << | Not Applicable | Completed | - | - |
| NCT00501540 | Neuroendocrine Tumors | Phase 2 | Completed | - | United States, Wisconsin ... more >> University of Wisconsin Paul P. Carbone Comprehensive Cancer Center Madison, Wisconsin, United States, 53792 Collapse << |
| NCT01091727 | Neurogenic Detrusor Overactivi... more >>ty Collapse << | Phase 3 | Completed | - | Canada, Ontario ... more >> Sunnybrook Health Sciences Centre Toronto, Ontario, Canada, M4N 3M5 Collapse << |
| NCT03201952 | Glucose Intolerance ... more >> Gastric Bypass Surgery Collapse << | Phase 1 | Recruiting | July 1, 2019 | United States, Tennessee ... more >> Vanderbilt University Medical Center Recruiting Nashville, Tennessee, United States, 37232 Contact: Michael Dole, MD 864-616-1389 michael.dole@vanderbilt.edu Collapse << |
| NCT02590549 | Keratoconus | Not Applicable | Completed | - | - |
| NCT00520468 | Myelodysplastic Syndrome | Phase 2 | Completed | - | United States, Texas ... more >> U.T.M.D. Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00520468 | - | - | Completed | - | - |
| NCT03483402 | Pseudoexfoliation Glaucoma ... more >> Primary Open-angle Glaucoma Collapse << | Not Applicable | Completed | - | - |
| NCT01916785 | Chronic Myelogenous Leukemia, ... more >>BCR/ABL Positive Collapse << | Phase 2 | Completed | - | Canada ... more >> Southern Alberta Cancer Research Institute Calgary, Canada Hôpital Charles LeMoyne Greenfield Park, Canada Queen elisabeth II Health Sciences Center Halifax, Canada CH Pierre LeGardeur Lachenaie, Canada Moncton City Hospital Moncton, Canada Hôpital Général Juif - Sir. Mortimer B. Davis Montréal, Canada Hôpital Maisonneuve-Rosemont Montréal, Canada Hôpital Royal Victoria Montréal, Canada Hôpital de l'Enfant Jésus - Centre hospitalier affilié universitaire de Québec Québec, Canada Pavillon Hôtel-Dieu de Québec - Centre hospitalier universitaire de Québec Québec, Canada France CHU Angers Angers, France Hôpital Avicenne Bobigny, France Institut Bergonie Bordeaux, France Hopital MORVAN Brest, France CH René Dubos Cergy Pontoise, France Hôpital d'Instruction de Armées Percy Clamart, France Hopital Henri MONDOR Creteil, France Hôpital Claude Huriez Lille, France CH Lyon Sud Lyon, France Institut Paoli-Calmettes Marseille, France Hôpital d'Annecy Metz Tessy, France C.H.U. Brabois Nancy, France CHU Hoptel dieu Nantes, France Hôpital l'Archet 1 Nice, France CHU Caremeau Nimes, France Hopital Saint Louis Paris, France Hôpital Necker-Enfants Malades Paris, France Hôpital St Antoine Paris, France CHU Poitiers Poitiers, France CHU Rennes - Pontchaillou Rennes, France Centre René Huguenin Saint Cloud, France Hôpital Purpan Toulouse, France CHRU Bretonneau Tours, France Central Hospital Versailles, France Collapse << |
| NCT01083108 | Obesity Baria... more >>tric Surgery Morbid Obesity Diabetes Mellitus Type 2 Diet Therapy Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> University of Maryland, Baltimore Baltimore, Maryland, United States, 21201-1595 National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 United States, Tennessee Vanderbilt University Nashville, Tennessee, United States, 37232 Collapse << |
| NCT02189889 | Anemia Cardio... more >>vascular Disease Collapse << | Phase 1 Phase 2 | Recruiting | June 2020 | United States, Texas ... more >> Clements University Hospital Recruiting Dallas, Texas, United States, 75390-8894 Contact: Kenni Landgraf, RN, BSN 214-645-8087 kenni.landgraf@utsouthwestern.edu Contact: Veronna Bell 214-645-8018 veronna.bell@utsouthwestern.edu Principal Investigator: Philip E. Greilich, MD Sub-Investigator: Michael Wait, MD Sub-Investigator: Eric Rosero, MD Sub-Investigator: Pietro Bajona, MD Sub-Investigator: Burner James, MD Clements University Hospital Recruiting Dallas, Texas, United States, 75390 Contact: Kenni Landgraf, RN, BSN 214-645-8087 kenni.landgraf@utsouthwestern.edu Collapse << |
| NCT02913170 | Hypercholesterolemia | Phase 4 | Completed | - | Korea, Republic of ... more >> Laboratory of Clinical Nutrigenetics/Nutrigenomics, Yonsei University. Seoul, Korea, Republic of, 120-749 Collapse << |
| NCT00404053 | Acute Coronary Syndrome | Phase 4 | Completed | - | - |
| NCT02923700 | Osteoarthritis | Phase 4 | Recruiting | December 2020 | Italy ... more >> Rizzoli Orthopaedic Institute Recruiting Bologna, Emilia Romagna, Italy, 40136 Contact: Elizaveta Kon, MD +39 0516366567 e.kon@biomec.ior.it Contact: Giuseppe Filardo, MD +39 0516366567 g.filardo@ior.it Collapse << |
| NCT00644657 | Coronary Artery Disease ... more >> Chest Pain Collapse << | Not Applicable | Completed | - | United States, Oklahoma ... more >> VA Medical Center, 1F187 Oklahoma City, Oklahoma, United States, 73104-5076 Collapse << |
| NCT00644657 | - | - | Completed | - | - |
| NCT01111864 | Bacterial Infections | Not Applicable | Completed | - | Kenya ... more >> Kikoneni Clinic Kikoneni, Kwale district, Kenya Collapse << |
| NCT02184637 | Malaria, Vivax | Phase 1 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT03413150 | - | - | Completed | - | - |
| NCT01149629 | Symptomatic Neurogenic Orthost... more >>atic Hypotension Collapse << | Phase 1 | Completed | - | United States, North Dakota ... more >> Cetero Research Fargo, North Dakota, United States, 58104 Collapse << |
| NCT02557685 | Clostridium Difficile Infectio... more >>n Collapse << | Phase 2 | Unknown | March 2017 | United States, Rhode Island ... more >> The Miriam Hospital Recruiting Providence, Rhode Island, United States, 02906 Contact: Colleen R. Kelly, MD 401-793-7080 ckelly2@lifespan.org Contact: Patrizia Curran, MD 401-793-7824 pcurran1@lifespan.org Principal Investigator: Colleen R. Kelly, MD Collapse << |
| NCT00320307 | HIV Infection | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site London, United Kingdom, SW10 9TH Collapse << |
| NCT02562014 | Obesity | Not Applicable | Completed | - | - |
| NCT03504774 | Food Allergy | Phase 2 | Not yet recruiting | March 10, 2026 | - |
| NCT01146301 | Carotid Stenosis | Phase 4 | Completed | - | Netherlands ... more >> St-Antonius Ziekenhuis Nieuwegein, Utrecht, Netherlands, 3435CM Collapse << |
| NCT01443169 | Healthy | Phase 1 | Completed | - | Belgium ... more >> Pfizer Investigational Site Bruxelles, Belgium, B-1070 Collapse << |
| NCT00882739 | Acute Myocardial Infarction | Phase 4 | Completed | - | Italy ... more >> Cardiovascular Department, Ospedale S.Donato Arezzo, AR, Italy, 52100 Collapse << |
| NCT00439140 | Overactive Bladder | Phase 3 | Terminated(In agreement with F... more >>DA the study was terminated based on data available.) Collapse << | - | United States, Pennsylvania ... more >> Philadelphia, Pennsylvania, United States Australia, Queensland Herston, Queensland, Australia Canada, British Columbia Victoria, British Columbia, Canada India Chennai, India Collapse << |
| NCT03362723 | Solid Tumors | Phase 1 | Active, not recruiting | March 29, 2019 | United States, Colorado ... more >> University of Colorado Aurora, Colorado, United States, 80045-2517 United States, Connecticut Yale Cancer Center; Medical Oncology New Haven, Connecticut, United States, 06520 United States, Missouri Washington University; Wash Uni. Sch. Of Med Saint Louis, Missouri, United States, 63110 United States, Nevada Comprehensive Cancer Centers of Nevada (CCCN) - Central Valley Las Vegas, Nevada, United States, 89169 United States, Oklahoma University of Oklahoma Health Sciences Center; Stephenson Cancer Center Oklahoma City, Oklahoma, United States, 73104 United States, Tennessee New Orleans Center for Clinical Research Knoxville, Tennessee, United States, 37920 United States, Texas Mary Crowley Medical Research Center Dallas, Texas, United States, 75230 Texas Oncology - Baylor Charles A. Sammons Cancer Center Dallas, Texas, United States, 75246 United States, Virginia Virginia Oncology Associates Norfolk, Virginia, United States, 23502 Canada, Ontario Princess Margaret Cancer Center Toronto, Ontario, Canada, M5G 1Z5 Canada, Quebec Jewish General Hospital / McGill University Montreal, Quebec, Canada, H3T 1E2 Collapse << |
| NCT00817999 | Healthy | Phase 4 | Completed | - | United States, Nebraska ... more >> Creighton Cardiac Center Omaha, Nebraska, United States, 68131 Collapse << |
| NCT03329001 | Solid Tumor | Phase 1 | Recruiting | December 2019 | United States, California ... more >> California Cancer Center for Research Excellence cCARE Recruiting Encinitas, California, United States, 92024 Principal Investigator: Alberto Bessudo, MD United States, Georgia Winship Cancer Institute of Emory University Recruiting Atlanta, Georgia, United States, 30322 Principal Investigator: R. Donald Harvey, PharmD United States, Tennessee Tennesse Oncology (SCRI) Recruiting Nashville, Tennessee, United States, 37203 Principal Investigator: Erika Hamilton, MD United States, Texas MD Anderson Recruiting Houston, Texas, United States, 77030 Principal Investigator: Sarina Piha-Paul, MD START- South Texas Accelerated Research Therapeutics, LLC Recruiting San Antonio, Texas, United States, 78229 Principal Investigator: Amita Patnaik, MD Collapse << |
| NCT02098174 | Glomerular Filtration Rate ... more >> Acute Kidney Injury Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> University of Maryland Baltimore, Maryland, United States, 21201 Collapse << |
| NCT01080612 | Healthy | Phase 1 | Completed | - | United States, Connecticut ... more >> Pfizer Investigational Site New Haven, Connecticut, United States, 06511 Collapse << |
| NCT03759067 | Angina, Stable | Not Applicable | Completed | - | - |
| NCT00631345 | Prediabetes O... more >>besity Collapse << | Not Applicable | Completed | - | United States, North Carolina ... more >> Wake Forest University School of Medicine Winston-Salem, North Carolina, United States, 27157 Collapse << |
| NCT01869465 | Schistosomiasis | Not Applicable | Unknown | June 2013 | Uganda ... more >> Primary schools Jinja, Uganda Collapse << |
| NCT00559260 | - | - | Completed | - | United States, Oklahoma ... more >> Oklahoma Foundation for Digestive Research Oklahoma City, Oklahoma, United States Collapse << |
| NCT01664702 | Childhood Obesity | Not Applicable | Completed | - | United States, Kansas ... more >> Energy Balance Lab, The University of Kansas Lawrence, Kansas, United States, 66045 Collapse << |
| NCT01575054 | Muscle Spasticity | Phase 3 | Completed | - | United States, California ... more >> Downey, California, United States United States, Florida Daytona Beach, Florida, United States Canada, Quebec Montreal, Quebec, Canada Czech Republic Prague, Czech Republic Germany Wiesbaden, Germany Würzburg, Germany Hungary Budapest, Hungary Korea, Republic of Gyeonggi-Do, Korea, Republic of Poland Krakow, Poland Russian Federation Krasnoyarsk, Russian Federation United Kingdom Stoke-on-Trent, England, United Kingdom Fazakerley, Liverpool, United Kingdom Collapse << |
| NCT01575054 | - | - | Completed | - | - |
| NCT00276198 | Iron Deficiency Anemia ... more >> Infectious Diseases Undernutrition Collapse << | Phase 3 | Completed | - | Israel ... more >> Ministry of Health Beer-Sheva, Israel, 10050 Collapse << |
| NCT00823290 | Hepatocellular Carcinoma | Phase 1 | Unknown | December 2010 | Germany ... more >> Department of Medicine 1, University Hospital Erlangen Recruiting Erlangen, Germany, 91054 Contact: Matthias Ocker, MD +49-6421-58 ext 68931 Matthias.Ocker@staff.uni-marburg.de Contact: Susanne Gahr, MD +49-9131-85 ext 35000 susanne.gahr@uk-erlangen.de Principal Investigator: Deike Strobel, MD Collapse << |
| NCT00284518 | Benign Prostatic Hyperplasia | Phase 2 | Completed | - | Australia ... more >> Murdoch, Australia Austria Vienna, Austria Canada, British Columbia Victoria, British Columbia, Canada Czech Republic Olomouc, Czech Republic France Paris Cedex 13, France Germany Braunschweig, Germany Italy Perugia, Italy Korea, Republic of Seoul, Korea, Republic of Slovakia Martin, Slovakia Taiwan Taipei, Taiwan United Kingdom London, United Kingdom Collapse << |
| NCT00284518 | - | - | Completed | - | - |
| NCT00821834 | Stable Angina ... more >> Myocardial Infarction Collapse << | Phase 3 | Completed | - | Japan ... more >> Sanofi-Aventis Administrative Office Tokyo, Japan Collapse << |
| NCT00834912 | Healthy | Phase 1 | Completed | - | - |
| NCT00501540 | - | - | Completed | - | - |
| NCT01901198 | Healthy Volunteers | Phase 1 | Completed | - | China, Beijing ... more >> Beijing YouAn Hospital, Capital Medical University Beijing, Beijing, China, 100069 Collapse << |
| NCT02004821 | Individuals With Constitutiona... more >>l Thinness Collapse << | Not Applicable | Completed | - | France ... more >> CHU de Saint-Étienne St-Etienne, France, 42055 Collapse << |
| NCT00439140 | - | - | Terminated(In agreement with F... more >>DA the study was terminated based on data available.) Collapse << | - | - |
| NCT01157000 | Healthy | Phase 1 | Completed | - | Belgium ... more >> Merksem, Belgium Collapse << |
| NCT00834912 | - | - | Completed | - | - |
| NCT03066401 | - | - | Terminated(insufficient inclus... more >>ion rate) Collapse << | - | France ... more >> Hôpital Roger Salengro, CHRU de Lille Lille, France Collapse << |
| NCT01703494 | Clostridium Difficile | Phase 2 | Unknown | September 2015 | United States, New York ... more >> Montefiore Medical Center The Bronx, New York, United States, 10467 United States, Rhode Island The Miriam Hospital Providence, Rhode Island, United States, 02906 Collapse << |
| NCT00876447 | Overactive Bladder | Phase 3 | Completed | - | - |
| NCT03066414 | - | - | Completed | - | France ... more >> Hôpital Roger Salengro, CHRU de Lille Lille, France Collapse << |
| NCT02910635 | Abnormalities, Cardiovascular | Phase 1 | Completed | - | United States, Texas ... more >> Covance Dallas, Texas, United States, 75247 Collapse << |
| NCT00876447 | - | - | Completed | - | - |
| 靶点 | Description | IC50 |
|---|---|---|
| HDAC | IC50:~1.8nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
